Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$100\ g$ of water is heated from $30^o\ C$ to $50^o\ C$ Ignoring the slight expansion of the water, the change in its internal energy is ...... $kJ$ (specific heat of water is $4184\ J/kg/K$)View Solution
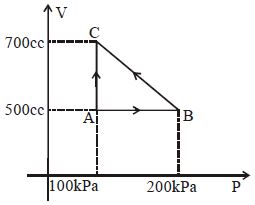
- 2A gas taken through cyclic process $ABCA$ is shown in figure. If $2.4\,cal.$ of heat is given in the process, what is value of $J$ ...... $J/cal$ (mechanical equivalent of heat)?View Solution
- 3$Assertion :$ Adiabatic expansion is always accompanied by fall in temperature.View Solution
$Reason :$ In adiabatic process, volume is inversely proportional to temperature. - 4View SolutionThe thermodynamic process, in which internal energy of the system remains constant is ...........
- 5View SolutionThe rate of recombination or generation are governed by the law(s) of
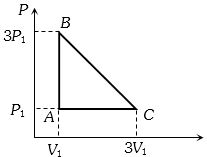
- 6An ideal gas is taken around the cycle $ABCA$ as shown in the $P-V $ diagram. The net work done by the gas during the cycle is equal toView Solution
- 7A mono atomic gas is supplied the heat $Q$ very slowly keeping the pressure constant. The work done by the gas will beView Solution
- 8$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope. - 9If heat given to a system is $6 \,kcal$ and work done is $ 6 kJ.$ Then change in internal energy is ....... $kJ$View Solution
- 10View SolutionTwo gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally, if the two gases are compressed to the same final volume
