An ideal gas at ${27^o}C$ is compressed adiabatically to $\frac{8}{{27}}$ of its original volume. If $\gamma = \frac{5}{3}$, then the rise in temperature is........ $K$
AIPMT 1999, Medium
(b) $\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^{\gamma - 1}} \Rightarrow {T_2} = 300\,{\left( {\frac{{27}}{8}} \right)^{\frac{5}{3} - 1}} = 300\,{\left( {\frac{{27}}{8}} \right)^{\frac{2}{3}}}$
$ = 300\,{\left\{ {{{\left( {\frac{{27}}{8}} \right)}^{1/3}}} \right\}^2} = 800\,{\left( {\frac{3}{2}} \right)^2} = 675K$
$ \Rightarrow \;\Delta T = 675 - 300 = 375\;K$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
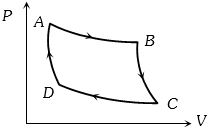
- 1The $P-V$ graph of an ideal gas cycle is shown here as below. The adiabatic process is described byView Solution
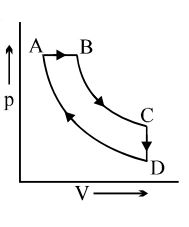
- 2A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
- 3This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1:$ In an adiabatic process, change in internal energy of a gas is equal to work done on/by the gas in the process.Statement $2 :$ The temperature of a gas remains constant in an adiabatic process.
- 4View SolutionWhen an ideal monoatomic gas is heated at constant pressure, fraction of heat energy supplied which increases the internal energy of gas, is
- 5View SolutionFirst law thermodynamics states that
- 6View SolutionWhich one is the correct option for the two different thermodynamic processes ?
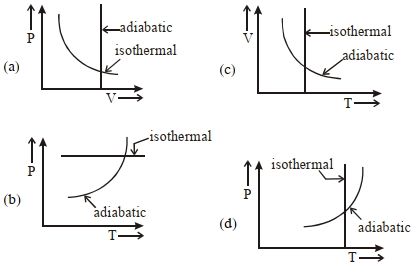
- 7View SolutionAir in a cylinder is suddenly compressed by a piston, which is then maintained at the same position. With the passage of time
- 8In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20\,J$ of heat and $8\,J$ of work is done on the gas. If the initial internal energy of the gas was $30\,J$. The final internal energy will be...... $J$View Solution
- 9View SolutionIn an isothermal expansion
- 10View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
