This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.
Statement $1:$ In an adiabatic process, change in internal energy of a gas is equal to work done on/by the gas in the process.
Statement $1:$ In an adiabatic process, change in internal energy of a gas is equal to work done on/by the gas in the process.
Statement $2 :$ The temperature of a gas remains constant in an adiabatic process.
AIEEE 2012, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A thermodynamic system is taken from state $A$ to $B$ along $ACB$ and is brought back to $A$ along $BDA$ as shown in the $PV$ diagram. The net work done during the complete cycle is given by the areaView Solution
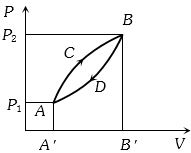
- 2An ideal gas expands from volume $V_1$ to $V_2$. This may be achieved by either of the three processes: isobaric, isothermal and adiabatic. Let $\Delta U$ be the change in internal energy of the gas, $Q$ be the quantity of heat added to the system and $W$ be the work done by the system on the gas. Identify which of the following statements is false for $\Delta U$?View Solution
- 3One mole of a monoatomic gas is carried along process $ABCDEA$ as shown in diagram. Find the net work done by gas ......... $J$View Solution
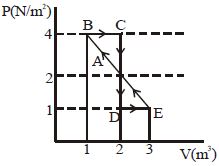
- 4View SolutionOut of the following which quantity does not depend on path
- 5An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
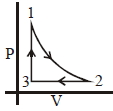
- 6Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
- 7One mole of a monatomic ideal gas is taken along two cyclic processes $E \rightarrow F \rightarrow G \rightarrow E$ and $E \rightarrow F \rightarrow H \rightarrow$ E as shown in the $PV$ diagram. The processes involved are purely isochoric, isobaric, isothermal or adiabatic. $Image$View Solution
Match the paths in List $I$ with the magnitudes of the work done in List $II$ and select the correct answer using the codes given below the lists.
List $I$ List $I$ $P.$ $\quad G \rightarrow E$ $1.$ $\quad 160 P_0 V_0 \ln 2$ $Q.$ $\quad G \rightarrow H$ $2.$ $\quad 36 P _0 V _0$ $R.$ $\quad F \rightarrow H$ $3.$ $\quad 24 P _0 V _0$ $S.$ $\quad F \rightarrow G$ $4.$ $\quad 31 P_0 V_0$ Codes: $ \quad \quad P \quad Q \quad R \quad S $
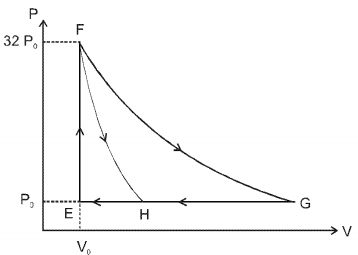
- 8An ideal gas is made to undergo a cycle depicted by the $p-V$ diagram given below. The curved line from $A$ to $B$ is an adiabat.Then,View Solution
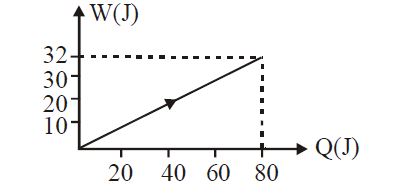
- 9View SolutionIn the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be
- 10A Carnot's engine works as a refrigerator between $250\, K$ and $300\, K$. It receives $500\, cal$ heat from the reservoir at the lower temperature. The amount of work done in each cycle to operate the refrigerator is ..... $J$View Solution
