Air in a cylinder is suddenly compressed by a piston, which is then maintained at the same position. With the passage of time
AIIMS 2000, Easy
(a) Due to compression the temperature of the system increases to a very high value.
This causes the flow of heat from system to the surroundings, thus decreasing the temperature.
This decrease in temperature results in decrease in pressure.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
$Reason :$ Entropy increases in all natural processes. - 2View SolutionWhich of the following can be coefficient of performance of refrigerator?
- 3View SolutionDuring an isothermal expansion of an ideal gas
- 4Half mole of an ideal monoatomic gas is heated at constant pressure of $1\, atm$ from $20\,^oC$ to $90\,^oC$. Work done by has is close to ..... $J$ (Gas constant $R = 8.31\, J/mol.K$)View Solution
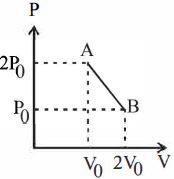
- 5'$n$' moles of an ideal gas undergoes a process $A \rightarrow B$ as shown in the figure. The maximum temperature of the gas during the process will beView Solution
- 6In an isothermal reversible expansion, if the volume of $96\, gm$ of oxygen at $27°C$ is increased from $70$ litres to $140$ litres, then the work done by the gas will beView Solution
- 7$V\, = \,K\,{\left( {\frac{P}{T}} \right)^{0.33}}$ where $k$ is constant. It is an,View Solution
- 8An insulator container contains $4\, moles$ of an ideal diatomic gas at temperature $T.$ Heat $Q$ is supplied to this gas, due to which $2 \,moles$ of the gas are dissociated into atoms but temperature of the gas remains constant. ThenView Solution
- 9A heat engine is involved with exchange of heat of $1915\, J,$ $-40\, J ,+125\, J$ and $-Q\,J$, during one cycle achieving an efficiency of $50.0 \%$. The value of $Q$ is.......$J$View Solution
- 10When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution