$Assertion :$ In adiabatic compression, the internal energy and temperature of the system get decreased.
$Reason :$ The adiabatic compression is a slow process.
$Reason :$ The adiabatic compression is a slow process.
AIIMS 2001, Easy
Adiabatic compression is a fast process. There is rise in temperature and also increase in internal energy. So, both are false.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn isothermic process, which statement is wrong
- 2A monoatomic gas is taken through a process $TP^{-1/3} =$ constant. If heat is given to the gasView Solution
- 3View SolutionIn isothermic process, which statement is wrong
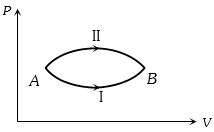
- 4A system goes from $A$ to $B$ via two processes $I$ and $II$ as shown in figure. If $\Delta {U_1}$ and $\Delta {U_2}$ are the changes in internal energies in the processes $I$ and $II$ respectively, thenView Solution
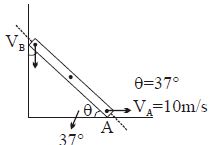
- 5Find $V_B = ?$View Solution
- 6View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
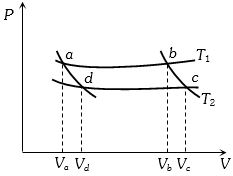
- 7In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
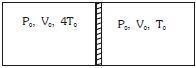
- 8Figure shows a cylindrical adiabatic container of total volume $2V_0$ divided into two equal parts by a conducting piston (which is free to move). Each part containing identical gas at pressure $P_0$ . Initially temperature of left and right part is $4T_0$ and $T_0$ respectively. An external force is applied on the piston to keep the piston at rest. Find the value of external force required when thermal equilibrium is reached. ( $A =$ Area of piston)View Solution
- 9A cyclic process $ABCD$ is shown in the given $P-V$ diagram. $P-T$ diagram that represents the same process isView Solution
- 10View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
