The state of a thermodynamic system is represented by
Easy
(c) The state of a system is defined by ascribing values to a sufficient number of variables. In mechanics, the state of a body is specified by means of a few measurable quantities, such as the position of centre of mass, its velocity and acceleration etc. In thermodynamics, however we disregard these mechanical coordinates and instead consider the interior of the system, find out directly or indirectly by experiments same measurable quantities upon which the internal state of the system for all thermodynamic purposes, the state of a system can be represented by specifying its pressure $p,$ volume $v$ and the temperature $T$ provided the system is in equilibrium.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe internal energy of an ideal gas depends upon
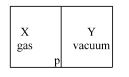
- 2A closed container is fully insulated from outside. One half of it is filled with an ideal gas $X$ separated by a plate Pfrom the other half $Y$ which contains a vacuum as shown in figure. When $P$ is removed, $X$ moves into $Y$. Which of the following statements is correct?View Solution
- 3View SolutionThe thermodynamic process, in which internal energy of the system remains constant is ...........
- 4View SolutionWhich of the following processes is reversible
- 5A Carnot engine, having an efficiency of $\eta = 1/10$ as heat engine, is used as a refrigerator. If the work done on the system is $10\ J$, the amount of energy absorbed from the reservoir at lower temperature is ....... $J$View Solution
- 6View SolutionWork done on or by a gas, in general depends upon the
- 7View SolutionWhen an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas, is
- 8In a given process for an ideal gas, $dW = 0$ and $dQ < 0.$ Then for the gasView Solution
- 9A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)View Solution
- 10View SolutionIf an ideal gas is compressed isothermally then
