An ideal gas goes from state $A$ to state $B$ via three different processes as indicated in the $P-V$ diagram. If $Q_1, Q_2, Q_3$ indicate the heat absorbed by the gas along the three processes and $\Delta U_1, \Delta U_2, \Delta U_3$
indicate the change in internal energy along the three processes respectively, then
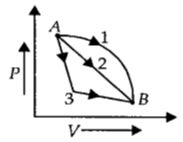
indicate the change in internal energy along the three processes respectively, then

AIPMT 2012, Medium
Change in internal energy is path independent and depends only on the initial and final states.
As the initial and final states in the three processes are same. Therefore,
$\Delta {u_1} = \Delta {u_2} = \Delta {u_3}$
Workdone, $W=Area\,under\,P-V\,graph$
As area under curve $1>$ area under curve $2>$ area under curve $3$
$\therefore {W_1} > {W_2} > {W_3}$
According to first law of thermodynamics,
$Q = W + \Delta u$
$As{W_1} > {W_2} > {W_3}\,and\,\Delta {u_1} = \Delta {u_2} = \Delta {u_3}$
$\therefore {Q_1} > {Q_2} > {Q_3}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The pressure $P_{1}$ and density $d_{1}$ of diatomic gas $\left(\gamma=\frac{7}{5}\right)$ changes suddenly to $P _{2}\left(> P _{1}\right)$ and $d _{2}$ respectively during an adiabatic process. The temperature of the gas increases and becomes $......$ times of its initial temperature.$\left(\right.$ given $\left.\frac{ d _{2}}{ d _{1}}=32\right)$View Solution
- 2View SolutionA gas is compressed adiabatically till its temperature is doubled. The ratio of its final volume to initial volume will be
- 3The value of $\eta$ may lie betweenView Solution
- 4View SolutionSelect the correct statement for work, heat and change in internal energy.
- 5A perfect gas of a given mass is heated first in a small vessel and then in a large vessel, such that their volumes remain unchanged. The $P-T$ curves areView Solution
- 6An ideal gas has volume ${V_0}$ at ${27^o}C.$ It is heated at constant pressure so that its volume becomes $2{V_0}.$ The final temperature isView Solution
- 7During an adiabatic process, if the pressure of a gas is found to be proportional to the cube of its absolute temperature, then the ratio of $\frac{C_p}{C_V}$ for the gas is:View Solution
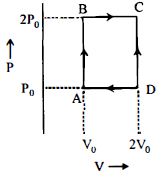
- 8An engine operates by taking $n\,moles$ of an ideal gas through the cycle $ABCDA$ shown in figure. The thermal efficiency of the engine is : (Take $C_v =1 .5\, R$, where $R$ is gas constant)View Solution
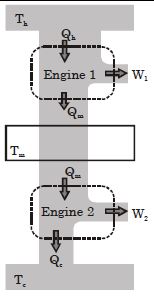
- 9Suppose that two heat engines are connected in series, such that the heat released by the first engine is used as the heat absorbed by the second engine, as shown in figure. The efficiencies of the engines are $\epsilon_1$ and $\epsilon_2$, respectively. The net efficiency of the combination is given by :View Solution
- 10View SolutionThe work done in which of the following processes is equal to the internal energy of the system?
