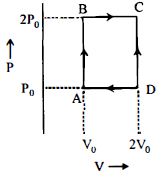
An engine operates by taking $n\,moles$ of an ideal gas through the cycle $ABCDA$ shown in figure. The thermal efficiency of the engine is : (Take $C_v =1 .5\, R$, where $R$ is gas constant)

JEE MAIN 2017, Diffcult
$Work - done\left( W \right) = {P_0}{V_0}$
According to principle of calorimetry
Heat given$ = {Q_{AB}} = {Q_{BC}}$
$ = n{C_V}d{T_{AB}} + n{C_p}d{T_{BC}}$
$ = \frac{3}{2}\left( {nR{T_B} - nR{T_A}} \right) + \frac{5}{2}\left( {nR{T_C} - nR{T_B}} \right)$
$ = \frac{3}{2}\left( {2{P_0}{V_0} - {P_0}{V_0}} \right) + \frac{5}{2}\left( {4{P_0}{V_0} - 2{P_{.0}}{V_0}} \right)$
$ = \frac{{13}}{2}{P_0}{V_0}$
Thermal efficiency of engine $\left( \eta \right)$
$ = \frac{W}{{{Q_{given}}}} = \frac{2}{{13}} = 0.15$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. The net work done on the gas in the cycle $ABCDA$ is ...... $R$View Solution

- 2In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
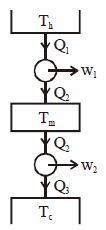
- 3Suppose that two heat engines are connected in series, such that the heat exhaust of the first engine is used as the heat input of the second engine as shown in figure. The efficiencies of the engines are $\eta_1$ and $\eta_2$, respectively. The net efficiency of the combination is given byView Solution
- 4View SolutionA gas is compressed adiabatically till its temperature is doubled. The ratio of its final volume to initial volume will be
- 5When the amount of work done is $333\, cal$ and change in internal energy is $167\, cal$, then the heat supplied is ....... $cal$View Solution
- 6A Carnot engine whose heat $\operatorname{sinks}$ at $27\,^{\circ} C$, has an efficiency of $25 \%$. By how many degrees should the temperature of the source be changed to increase the efficiency by $100 \%$ of the original efficiency $?$View Solution
- 7A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
- 8Two Carnot engines $A$ and $B$ are operated in series. The first one, $A,$ receives heat at $T_1(= 600\,K)$ and rejects to a reservoir at temperature $T_2.$ The second engine $B$ receives heat rejected by the first engine and, in turns, rejects to a heat reservoir at $T_3 (=400\,K).$ Calculate the temperature $T_2$ if the work outputs of the two engines are equal ..... $K$View Solution
- 9In an adiabatic process, the state of a gas is changed from ${P_1},{V_1},{T_1} $ to ${P_2},{V_2},{T_2}$. Which of the following relation is correctView Solution
- 10View SolutionIf a system undergoes contraction of volume then the work done by the system will be
