An ideal gas, initially in state $\left( P _{12}, V _1, T _1\right)$ is expanded isobarically to $\left( P _{12}, V _2, T _2\right)$, then adiabatically $\left( P _{34}, V _3, T _3\right)$. It is then contracted isobarically to $\left( P _{34}, V _4, T _4\right)$ and finally adiabatically back to the initial state. The efficiency of this cycle is
KVPY 2021, Advanced
(A)
$P _{12} V _2^\gamma= P _{34} V_3^\gamma \quad \dots(i)$
$P _{34} V_4^\gamma= P _{12} V_1^\gamma \quad \dots(ii)$
Multiply $(i)$ and $(ii)$
$V _2^\gamma \cdot V _4^\gamma= V _1^\gamma \cdot V _3^\gamma$
$V _2 V _4= V _1 V _3 \Rightarrow\left(\frac{ V _2}{ V _3}=\frac{ V _1}{ V _4}\right)= K _4$
$\eta=\frac{\text { Work done }}{ Q _{\text {suppl. }}}=1-\frac{ Q _{\text {rej }}}{ Q _{\text {supp }}}=1-\frac{ n _{ C / P } \Delta T _{34}}{ n _{ C / P } \Delta T _{12}}$
$=1-\frac{\Delta( PV )_{34}}{\Delta( PV )_{12}}=1-\frac{ P _{34}\left( V _3- V _4\right)}{ P _{12}\left( V _2- V _1\right)}$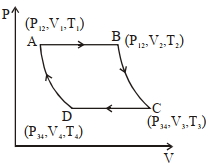
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ The heat supplied to a system is always equal to the increase in its internal energy.View Solution
$Reason :$ When a system changes from one thermal equilibrium to another, some heat is absorbed by it. - 2Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative.View Solution
- 3A tyre filled with air $({27^o}C,$ and $2$ atm) bursts, then what is temperature of air ....... $^oC$ $(\gamma = 1.5)$View Solution
- 4$N$ moles of an ideal diatomic gas are in a cylinder at temperature $T$. suppose on supplying heat to the gas, its temperature remain constant but $n$ moles get dissociated into atoms. Heat supplied to the gas isView Solution
- 5Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at $T_1 = 800\, K$ and rejects to sink at $T_2K$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at $T_3 = 300\, K$. If the work outputs of two engines are equal, then the value of $T_2$ is ...... $K$View Solution
- 6View SolutionWhen heat in given to a gas in an isobaric process, then
- 7Three moles of an ideal gas $\left( {{C_P} = \frac{7}{2}R} \right)$ at pressure ${P_A}$ and temperature ${T_A}0$ is isothermally expanded to twice its initial volume. It is then compressed at constant pressure to its original volume. Finally the gas is compressed at constant volume to its original pressure ${P_A}.$ The correct $P-V$ and $P-T$ diagrams indicating the process areView Solution
- 8The temperature inside and outside a refrigerator are $273 \,K$ and $300 \,K$ respectively. Assuming that the refrigerator cycle is reversible, for every joule of work done, the heat delivered to the surrounding will be nearly ........ $J$View Solution
- 9$List I$ describes thermodynamic processes in four different systems. $List II$ gives the magnitudes (either exactly or as a close approximation) of possible changes in the internal energy of the system due to the process.View Solution
$List-I$ $List-II$ ($I$) $10^{-3} kg$ of water at $100^{\circ} C$ is converted to steam at the same temperature, at a pressure of $10^5 Pa$. The volume of the system changes from $10^{-6} m ^3$ to $10^{-3} m ^3$ in the process. Latent heat of water $=2250 kJ / kg$. ($P$) $2 kJ$ ($II$) $0.2$ moles of a rigid diatomic ideal gas with volume $V$ at temperature $500 K$ undergoes an isobaric expansion to volume $3 V$. Assume $R=8.0 Jmol ^1 K^{-1}$. ($Q$) $7 kJ$ ($III$) On mole of a monatomic ideal gas is compressed adiabatically from volume $V=\frac{1}{3} m^3$ and pressure $2 kPa$ to volume $\frac{v}{8}$ ($R$) $4 kJ$ ($IV$) Three moles of a diatomic ideal gas whose molecules can vibrate, is given $9 kJ$ of heat and undergoes isobaric expansion. ($S$) $5 kJ$ ($T$) $3 kJ$ Which one of the following options is correct?
- 10View SolutionAir in a cylinder is suddenly compressed by a piston, which is then maintained at the same position. With the passage of time
