$List I$ describes thermodynamic processes in four different systems. $List II$ gives the magnitudes (either exactly or as a close approximation) of possible changes in the internal energy of the system due to the process.
| $List-I$ | $List-II$ |
| ($I$) $10^{-3} kg$ of water at $100^{\circ} C$ is converted to steam at the same temperature, at a pressure of $10^5 Pa$. The volume of the system changes from $10^{-6} m ^3$ to $10^{-3} m ^3$ in the process. Latent heat of water $=2250 kJ / kg$. | ($P$) $2 kJ$ |
| ($II$) $0.2$ moles of a rigid diatomic ideal gas with volume $V$ at temperature $500 K$ undergoes an isobaric expansion to volume $3 V$. Assume $R=8.0 Jmol ^1 K^{-1}$. | ($Q$) $7 kJ$ |
| ($III$) On mole of a monatomic ideal gas is compressed adiabatically from volume $V=\frac{1}{3} m^3$ and pressure $2 kPa$ to volume $\frac{v}{8}$ | ($R$) $4 kJ$ |
| ($IV$) Three moles of a diatomic ideal gas whose molecules can vibrate, is given $9 kJ$ of heat and undergoes isobaric expansion. | ($S$) $5 kJ$ |
| ($T$) $3 kJ$ |
Which one of the following options is correct?
IIT 2022, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn the adiabatic compression, the decrease in volume is associated with
- 2The initial pressure and volume of an ideal gas are $P_0$ and $V_0$. The final pressure of the gas when the gas is suddenly compressed to volume $\frac{ V _0}{4}$ will be (Given $\gamma=$ ratio of specific heats at constant pressure and at constant volume)View Solution
- 3The pressure and volume of a gas are changed as shown in the $P-V$ diagram in this figure. The temperature of the gas will ........View Solution
- 4Consider two containers $A$ and $B$ containing identical gases at the same pressure, volume and temperature. The gas in container $A$ is compressed to half of its original volume isothermally while the gas in container $B$ is compressed to half of its original value adiabatically. The ratio of final pressure of gas in $B$ to that of gas in $A$ isView Solution
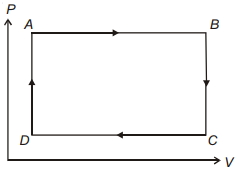
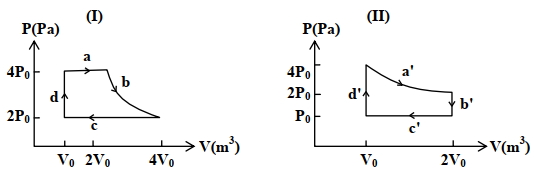
- 5One mole of an ideal gas undergoes two different cyclic processes I and II, as shown in the $P-V$ diagrams below. In cycle I, processes $a, b, c$ and $d$ are isobaric, isothermal, isobaric and isochoric, respectively. In cycle II, processes $a^{\prime}, b^{\prime}, c^{\prime}$ and $d^{\prime}$ are isothermal, isochoric, isobaric and isochoric, respectively. The total work done during cycle I is $W_I$ and that during cycle II is $W_{I I}$. The ratio $W_I / W_{I I}$ is . . . .View Solution
(image)
- 6A thermodynamic cycle takes in heat energy at a high temperature and rejects energy at a lower temperature. If the amount of energy rejected at the low temperature is $3$ times the amount of work done by the cycle, the efficiency of the cycle isView Solution
- 7An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?View Solution
- 8View SolutionIn thermodynamic processes which of the following statements is not true?
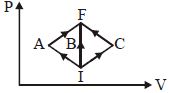
- 9In the $P-V$ diagram, $I$ is the initial state and $F$ is the final state. The gas goes from $I$ to $F$ by $(i)\,IAF,\,\,(ii)\,IBF,\,\,(iii) ICF$. The heat absorbed by the gas isView Solution
- 10Jet aircrafts fly at altitudes above $30000 \,ft$, where the air is very cold at $-40^{\circ} C$ and the pressure is $0.28 \,atm$. The cabin is maintained at $1 \,atm$ pressure by means of a compressor which exchanges air from outside adiabatically. In order to have a comfortable cabin temperature of $25^{\circ} C$, we will require in additionView Solution
