An ideal gas is filled in a vessel, then
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ At a given temperature the specific heat of a gas at constant pressure. is always greater than its specific heat at constant volumeView Solution
$Reason :$ When a gas is heated at constant volume some extra heat is needed compared to that at constant pressure for doing work in expansion.
- 2View SolutionMean kinetic energy per degree of freedom of gas molecules is
- 3View SolutionA gas is filled in the cylinder shown in the figure. The two pistons are joined by a string. If the gas is heated, the pistons will
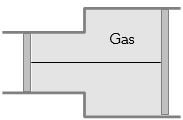
- 4The $r.m.s.$ velocity will be greater forView Solution
- 5A diatomic gas follows equation $PV^m =$ constant, during a process. What should be the value of $m$ such that its molar heat capacity during process $= R$View Solution
- 6A mixture of hydrogen and oxygen has volume $2000 \; cm ^{3}$, temperature $300 \; K$, pressure $100 \; kPa$ and mass $0.76 \; g$ The ratio of number of moles of hydrogen to number of moles of oxygen in the mixture will beView Solution
- 7A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $1.4$. Vessel is moving with speed $v$ and is suddenly brought to rest. Assuming no heat is lost to the surrounding and vessel temperature of the gas increases by ... ( $R =$ universal gas constant )View Solution
- 8From the following $P-T$ graph what interference can be drawnView Solution
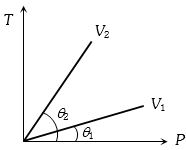
- 9A very tall vertical cylinder is filled with a gas of molar mass $M$ under isothermal conditions at temperature $T.$ The density and pressure of the gas at the base of the container is $\rho_0$ and $p_0$, respectively Choose the correct statement $(s)$ if gravity is assumed to be constant throughout the containerView Solution
- 10Two closed containers of equal volume filled with air at pressure $P_0$ and temperature $T_0$. Both are connected by a narrow tube. If one of the container is maintained at temperature $T_0$ and other at temperature $T$, then new pressure in the containers will beView Solution
