A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $1.4$. Vessel is moving with speed $v$ and is suddenly brought to rest. Assuming no heat is lost to the surrounding and vessel temperature of the gas increases by ... ( $R =$ universal gas constant )
JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionAn ideal gas is filled in a vessel, then
- 2View SolutionThe time average of the kinetic energy of one molecule of a gas taken over a long period of time
- 3A container is divided into two chambers by a partition. The volume of first chamber is $4.5$ litre and second chamber is $5.5$ litre. The first chamber contain $3.0$ moles of gas at pressure $2.0\, atm$ and second chamber contain $4.0$ moles of gas at pressure $3.0\, atm$ .After the partition is removed and the mixture attains equilibrium, then, the common equilibrium pressure existing in the mixture is $x \times 10^{-1} atm$. Value of $x$ is.........View Solution
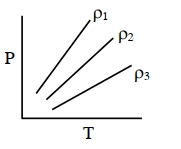
- 4$P-T$ diagram of an ideal gas having three different densities $\rho_1, \rho_2, \rho_3$ (in three different cases) is shown in the figure. Which of the following is correct :View Solution
- 5$E_0$ and $E_H$ respectively represents the average kinetic energy of a molecule of oxygen and hydrogen. If the two gases are at the same temperature, which of the following statement is true?View Solution
- 6View SolutionSelect the incorrect statement about the specific heats of a gaseous system.
- 7If the r.m.s. speed of chlorine molecule is $490\,m / s$ at $27^{\circ}\,C$, the r.m.s. speed of argon molecules at the same temperature will be $......\,m/s$ (Atomic mass of argon $=39.9\,u$, molecular mass of chlorine $=70.9\,u )$View Solution
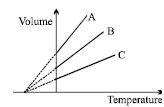
- 8The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $B$. Then the expansion of the same ideal gas of mass $2\, m$ at a pressure $2\,P$ is given by the straight lineView Solution
- 9The ratio of specific heats $(\gamma )$ of a ideal gas is given byView Solution
- 10A gas is filled in a cylinder, its temperature is increased by $20\%$ on kelvin scale and volume is reduced by $10\%$ . How much percentage of the gas will leak out at constant pressure ...... $\%$View Solution
