A mixture of hydrogen and oxygen has volume $2000 \; cm ^{3}$, temperature $300 \; K$, pressure $100 \; kPa$ and mass $0.76 \; g$ The ratio of number of moles of hydrogen to number of moles of oxygen in the mixture will be
JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$125\, ml$ of gas $A$ at $0.60$ atmosphere and $150\, ml$ of gas $B$ at $0.80$ atmosphere pressure at same temperature is filled in a vessel of $1$ litre volume. What will be the total pressure of mixture at the same temperature ............... $\mathrm{atmosphere}$View Solution
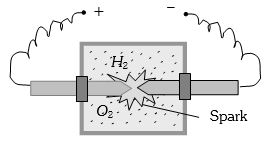
- 2A vessel contains $14 \,gm \,(7 $ moles) of hydrogen and $96\, gm$ ($9$ moles) of oxygen at $STP.$ Chemical reaction is induced by passing electric spark in the vessel till one of the gases is consumed. The temperature is brought back to it's starting value $273 K.$ The pressure in the vessel is ...... $atm$View Solution
- 3View SolutionThe absolute zero is the temperature at which
- 4A polyatomic gas with $n$ degrees of freedom has a mean energy per molecule given byView Solution
($N$ is Avogadro's number)
- 5The average kinetic energy of a helium atom at ${30^o}C$ isView Solution
- 6$C{O_2}(O - C - O)$ is a triatomic gas. Mean kinetic energy of one gram gas will be (If $N-$Avogadro's number, $k-$Boltzmann's constant and molecular weight of $C{O_2} = 44$)View Solution
- 7View SolutionYou have two containers of equal volume. One is full of helium gas. The other holds an equal mass of nitrogen gas. Both gases have the same pressure. How does the temperature of the helium compare to the temperature of the nitrogen ?
- 8When the temperature of a gas is raised from $27^o C$ to $90^o C$, the percentage increase in the $r.m.s.$ velocity of the molecules will be ..... $\%$View Solution
- 9The equation of state corresponding to $8 \,g$ of ${O_2}$ isView Solution
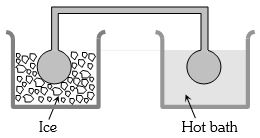
- 10Two identical glass bulbs are interconnected by a thin glass tube. A gas is filled in these bulbs at $N.T.P.$ If one bulb is placed in ice and another bulb is placed in hot bath, then the pressure of the gas becomes $1.5$ times. The temperature of hot bath will be ....... $^oC$View Solution