An ideal gas is subjected to a thermodynamic process $PV^{2.5} = 0.40$ where $P$ is in $Pa$ and $V$ is in $m^3$.What is the slope of the $P-V$ curve with volume plotted against $x-$ axis at $V=1\, m^3$ ?
Medium
$\mathrm{PV}^{2.5}=0.40$
$\mathrm{P}=0.40 \mathrm{V}^{-2.5}$
$\frac{\mathrm{dP}}{\mathrm{dV}}=0.40\left[-2.5 \mathrm{V}^{-3.5}\right]$
at $\quad V=1$
$\left(\frac{\mathrm{dP}}{\mathrm{dV}}\right)_{\mathrm{V}=1}=-1$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
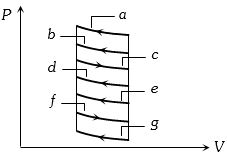
- 1The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done on the system in going from $A → B → C$ is $50 J$ and $ 20\, cal$ heat is given to the system. The change in internal energy between $A$ and $C$ is ...... $J$View Solution
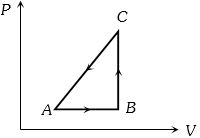
- 2One mole of an ideal gas in initial state $\mathrm{A}$ undergoes a cyclic process $A B C A$, as shown in the figure. Its pressure at $A$ is $\mathrm{P}_0$. Choose the correct option$(s)$ from the followingView Solution
$(A)$ Internal energies at $\mathrm{A}$ and $\mathrm{B}$ are the same
$(B)$ Work done by the gas in process $\mathrm{AB}$ is $\mathrm{P}_0 \mathrm{~V}_0 \ln 4$
$(C)$ Pressure at $C$ is $\frac{P_0}{4}$
$(D)$ Temperature at $\mathrm{C}$ is $\frac{\mathrm{T}_0}{4}$
- 3$Assertion :$ Adiabatic expansion is always accompanied by fall in temperature.View Solution
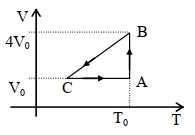
$Reason :$ In adiabatic process, volume is inversely proportional to temperature. - 4An ideal heat engine working between temperature $T_1$ and $T_2 $ has an efficiency $\eta$, the new efficiency if both the source and sink temperature are doubled, will beView Solution
- 5A Carnot engine whose sink is at $300\, K$ has an efficiency of $40\%.$ By how much should the temperature of source be increased so as to increase its efficiency by $50\%$ of original efficiency ..... $K$View Solution
- 6View SolutionTemperature is a measurement of coldness or hotness of an object. This definition is based on
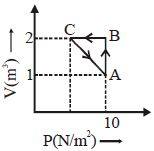
- 7An ideal gas is taken through the cycle $A \to B \to C \to A,$ as shown in the figure. What is the change in internal energy ...... $J$.View Solution
- 8View SolutionOut of the following which quantity does not depend on path
- 9A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
- 10The $P-V$ diagram shows seven curved paths (connected by vertical paths) that can be followed by a gas. Which two of them should be parts of a closed cycle if the net work done by the gas is to be at its maximum valueView Solution