A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energy
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
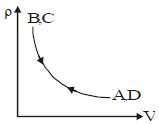
- 1Density $vs$ volume graph is shown in the figure. Find corresponding pressure $vs$ temperature graphView Solution
- 2View SolutionWork done per mol in an isothermal change is
- 3An ideal gas undergoes an adiabatic process obeying the relation $PV^{4/3} =$ constant. If its initial temperature is $300\,\, K$ and then its pressure is increased upto four times its initial value, then the final temperature is (in Kelvin):View Solution
- 4View SolutionIn an isothermal expansion
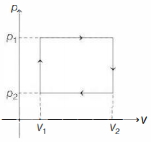
- 5$n$ moles of a van der Waals' gas obeying the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$, where $a$ and $b$ are gas dependent constants, is made to undergo a cyclic process that is depicted by a rectangle in the $p-V$ diagram as shown below. What is the heat absorbed by the gas in one cycle?View Solution
- 6View SolutionThe adiabatic Bulk modulus of a perfect gas at pressure is given by
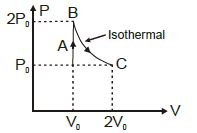
- 7A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution
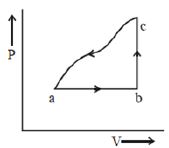
- 8A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
- 9View SolutionA container that suits the occurrence of an isothermal process should be made of
- 10A diatomic gas $(\gamma=1.4)$ does $200 \mathrm{~J}$ of work when it is expanded isobarically. The heat given to the gas in the process is:View Solution
