A Carnot engine whose sink is at $300\, K$ has an efficiency of $40\%.$ By how much should the temperature of source be increased so as to increase its efficiency by $50\%$ of original efficiency ..... $K$
AIPMT 2006, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$100\ g$ of water is heated from $30^o C$ to $50^o C$. Ignoring the slight expansion of the water, the change in its internal energy is .......$kJ$ (specific heat of water is $4184\ J/kg/K$):View Solution
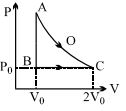
- 2An ideal gas is taken from point $A$ to point $C$ on $P-V$ diagram through two process $AOC$ and $ABC$ as shown in the figure. Process $AOC$ is isothermalView Solution
- 3A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an abosolute temperature $T$ and rejects heat to a sink at a temperature of $T/3.$ The amount of heat rejected isView Solution
- 4View SolutionThe work done in which of the following processes is zero
- 5View SolutionFirst law of thermnodynamics is given by
- 6A Carnot engine works first between $200^{\circ} C$ and $0^{\circ} C$ and then between $0^{\circ} C$ and $-200^{\circ} C$. The ratio of its efficiency in the two cases isView Solution
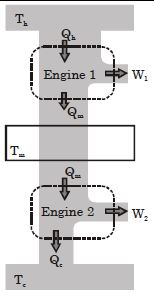
- 7Suppose that two heat engines are connected in series, such that the heat released by the first engine is used as the heat absorbed by the second engine, as shown in figure. The efficiencies of the engines are $\in_1$ and $\in_2$, respectively. The net efficiency of the combination is given by :View Solution
- 8View SolutionFor an isometric process
- 9One mole of a monatomic ideal gas undergoes an adiabatic expansion in which its volume becomes eight times its initial value. If the initial temperature of the gas is $100 K$ and the universal gas constant $R =8.0 Jmol ^{-1} K ^{-1}$, the decrease in its internal energy, in Joule, is. . . . .View Solution
- 10The efficiency of Carnot's engine operating between reservoirs, maintained at temperatures ${27^o}C$ and $ - {123^o}C,$ is ...... $\%$View Solution
