An ideal gas is taken through the cycle $A \to B \to C \to A,$ as shown in the figure. What is the change in internal energy ...... $J$.
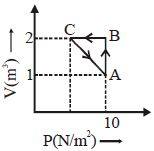

Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionTwo gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally
- 2One mole of a gas expands obeying the relation as shown in the $P/V$ diagram. The maximum temperature in this process is equal toView Solution
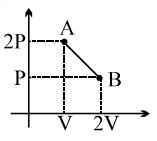
- 3View SolutionIf a system undergoes contraction of volume then the work done by the system will be
- 4A bubble from bottom of lake rises to its surface. Its volume doubles in the process. Assuming isothermal conditions, atmospheric pressure $= 75\, cm$ of $Hg$ and ratio of densities of mercury and water $40/3$. The depth of lake will be ..... $m$View Solution
- 5One mole of an ideal gas undergoes a cyclic process, consisting of two isochores and two isobars. Temperature at $1$ and $3$ equal to $T_1$ and $T_3$ respectively. The work done by the gas over the cycle, if the point $2$ and $4$ lie on the same isothermView Solution
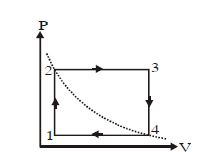
- 6View SolutionWhen heat in given to a gas in an isobaric process, then
- 7Two samples $A$ and $B$ of a gas initially at the same pressure and temperature are compressed from volume $ V$ to $ V/2$ ($A$ isothermally and adiabatically). The final pressure of $ A$ isView Solution
- 8A perfect gas of a given mass is heated first in a small vessel and then in a large vessel, such that their volumes remain unchanged. The $P-T$ curves areView Solution
- 9The adiabatic elasticity of hydrogen gas $(\gamma = 1.4)$ at $NTP$ isView Solution
- 10The efficiency of Carnot engine when source temperature is $T_1$ and sink temperature is $T_2$ will beView Solution
