Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A bubble from bottom of lake rises to its surface. Its volume doubles in the process. Assuming isothermal conditions, atmospheric pressure $= 75\, cm$ of $Hg$ and ratio of densities of mercury and water $40/3$. The depth of lake will be ..... $m$View Solution
- 2A rigid diatomic ideal gas undergoes an adiabatic process at room temperature. The rational between temperature and volume for the process is $TV^x =$ constant, then $x$ isView Solution
- 3View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
- 4View SolutionIn an isothermal change, an ideal gas obeys
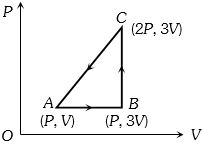
- 5An ideal gas with constant heat capacity $C_V=\frac{3}{2} n R$ is made to carry out a cycle that is depicted by a triangle in the figure given below. The following statement is true about the cycle.View Solution
- 6View SolutionThe efficiency of a Carnot engine depends upon
- 7A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
- 8In an adiabatic change, the pressure $P$ and temperature $T$ of a monoatomic gas are related by the relation $P \propto {T^C}$, where $C$ equalsView Solution
- 9$Assertion :$ Thermodynamic process in nature are irreversible.View Solution
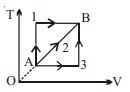
$Reason :$ Dissipative effects cannot be eliminated. - 10A given mass of a gas expands from a state $A$ to the state $B$ by three paths $1, 2$ and $3$ as shown in $T-V$ indicator diagram. If $W_1, W_2$ and $W_3$ respectively be the work done by the gas along the three paths, thenView Solution