A bubble from bottom of lake rises to its surface. Its volume doubles in the process. Assuming isothermal conditions, atmospheric pressure $= 75\, cm$ of $Hg$ and ratio of densities of mercury and water $40/3$. The depth of lake will be ..... $m$
Medium
Isothermal condition $\mathrm{P}_{1} \mathrm{V}_{1}=\mathrm{P}_{2} \mathrm{V}_{2}$
$\left(\rho_{1} {g} 5\right)(2 \mathrm{V})=\left[\left(\rho_{1} g 75\right)+\rho_{2}{g} h\right] V$
$\Rightarrow \rho_{1} \mathrm{g} 75=\rho_{2} {g} \mathrm{h}$
$\Rightarrow \mathrm{h}=75 \times \frac{\rho_{1}}{\rho_{2}}=\frac{75 \times 40}{3}=1000 \mathrm{cm}=10 \mathrm{m}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Match List$-I$ with List$-II$View Solution
List$-I$ List$-II$ $(a)$ Isothermal $(i)$ Pressure constant $(b)$ Isochoric $(ii)$ Temperature constant $(c)$ Adiabatic $(iii)$ Volume constant $(d)$ Isobaric $(iv)$ Heat content is constant Choose the correct answer from the options given below
- 2View SolutionA cycle tyre bursts suddenly. This represents an
- 3View SolutionFirst law thermodynamics states that
- 4$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
- 5A Carnot engine takes $3 \times {10^6}\,cal$. of heat from a reservoir at $627°C$, and gives it to a sink at $27°C.$ The work done by the engine isView Solution
- 6Initial pressure and volume of a gas are $P$ and $V$ respectively. First it is expanded isothermally to volume $4V$ and then compressed adiabatically to volume $V$ . The final pressure of gas will be (given $\gamma = 3/2$ )View Solution
- 7View SolutionIn the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be
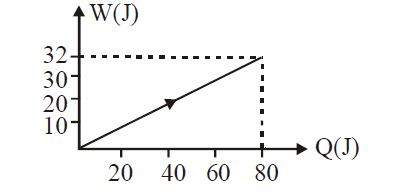
- 8View SolutionIn the cyclic process shown in the figure, the work done by the gas in one cycle is
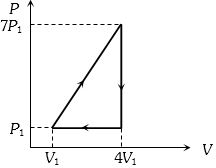
- 9A $P-T$ graph is shown for a cyclic process. Select correct statement regarding thisView Solution
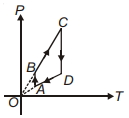
- 10A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion isView Solution
