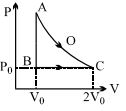
An ideal gas is taken from point $A$ to point $C$ on $P-V$ diagram through two process $AOC$ and $ABC$ as shown in the figure. Process $AOC$ is isothermal

Advanced
Being a state-function, change in in internal energy$(\Delta U)$ is the same for the two processes AOC and $ABC,$ i.e. $\Delta U_{A O C}=\Delta U_{A B C}$
Work done $=W=$ area under $P-V$ diagram Hence $W_{A O C}>W_{A B C}$
$\Rightarrow \Delta U_{A O C}+W_{A O C}>\Delta U_{A B C}+W_{A B C}$
$\Rightarrow \Delta Q_{A O C}>\Delta Q_{A B C}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $8.0 \,m ^{3}$ to $1.0 \,m ^{3}$.
Step $2$ Then expanded isothermally at temperature $T_{1}$ to volume $10.0 \,m ^{3}$.
Step $3$ Then expanded adiabatically to volume $80.0 \,m ^{3}$.
Step $4$ Then compressed isothermally at temperature $T_{2}$ to volume $8.0 \,m ^{3}$.
Then, $T_{1} / T_{2}$ is
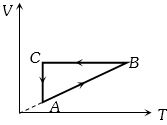
- 2A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
- 3In an Isothermal change, the change in pressure and volume of a gas can be represented for three different temperature; $T _3 > T _2 > T _1$ as :View Solution
- 4In an adiabatic process, the density of a diatomic gas becomes $32$ times its initial value. The final pressure of the gas is found to be $n$ times the initial pressure. The value of $n$ isView Solution
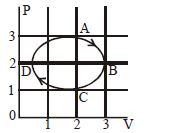
- 5The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA.$ The part $ABC$ is a semicircle and $CDA$ is half of an ellipse. Then,View Solution
- 6An ideal gas is expanding such that $\mathrm{PT}^2=$ constant. The coefficient of volume expansion of the gas isView Solution
- 7A certain amount of gas is taken through a cyclic process $(A\,B\,C\,D\,A)$ that has two isobars, one isochore and one isothermal. The cycle can be represented on a $P-V$ indicator diagram asView Solution
- 8Two Carnot engines $A$ and $B$ are operated in series. Engine $A$ receives heat from a reservoir at $600\,K$ and rejects heat to a reservoir at temperature $T$. Engine $B$ receives; heat rejected by engine $A$ and in turn rejects it to a reservoir at $100\,K$. If the efficiencies of the two engines $A$ and $B$ are represented by ${\eta _A}$ and ${\eta _B}$ respectively, then what is the value of $\frac{{{\eta _A}}}{{{\eta _B}}}$View Solution
- 9A balloon filled with helium $\left(32^{\circ} C \right.$ and $1.7\; atm$.) bursts. Immediately afterwards the expansion of helium can be considered asView Solution
- 10View SolutionIn an isothermal expansion
