Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$View Solution
- 2View SolutionA perfect gas contained in a cylinder is kept in vacuum. If the cylinder suddenly bursts, then the temperature of the gas
- 3One mole of an ideal gas is taken through an adiabatic process where the temperature rises from $27^{\circ} {C}$ to $37^{\circ} {C}$. If the ideal gas is composed of polyatomic molecule that has $4$ vibrational modes which of the following is true?View Solution
- 4When a system is taken from state $i$ to a state $f$ along path $iaf, \,Q = 50\,J$ and $W = 20J.$ Along path $ibf, \,Q = 35J.$ If $W = - 13J$ for the curved return path $f i, Q$ for this path is ...... $J$View Solution
- 5This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1:$ In an adiabatic process, change in internal energy of a gas is equal to work done on/by the gas in the process.Statement $2 :$ The temperature of a gas remains constant in an adiabatic process.
- 6View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
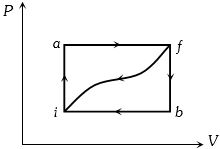
- 7View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
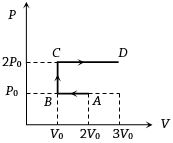
- 8$P-V$ diagram of an ideal gas is as shown in figure. Work done by the gas in process $ABCD$ isView Solution
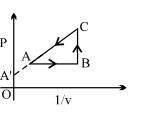
- 9Figure shows, the adiabatic curve on a $\log T$ and log $V$ scale performed on ideal gas. The gas is ............View Solution
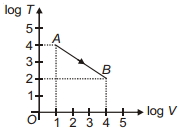
- 10The adiabatic elasticity of hydrogen gas $(\gamma = 1.4)$ at $NTP$ isView Solution