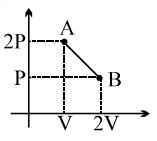
and minima.
since the graph shown here is a straight line, we can write it's equation in the form of $:$
$y=m x+c$
here $y=P, x=V, m=\frac{P_{2}-P_{1}}{V_{2}-V_{1}}=\frac{\frac{P_{0}}{2}-P_{0}}{2 V_{0}-V_{0}}$
$\Rightarrow m=\frac{-P_{0}}{2 V_{0}}$
$c$ is the intercept and $c=\frac{3 P_{0}}{2}$
We can find $c$ by extra polating the graph Now
$P=\frac{-P_{0}}{2 V_{0}} V+\frac{3 P_{0}}{2}$ $...(1)$
Also, from equation of state, we know the $P V=n R T$
for $1$ mole $P V=R T$
or $P=\frac{R T}{V}$
substituting this in equation $( 1)$
$\frac{R T}{V}=\frac{-P_{0}}{2 V_{0}} V+\frac{3 P_{0}}{2}$
$T=\frac{-\bar{P}_{0}}{2 R V_{0}} \cdot V^{2}+\frac{3 P_{0}}{2 R} V$
To find the minimum temperature, we will differentiate the above equation
i.e., $\frac{d T}{d V}=-\frac{2 P_{0}}{2 R V_{0}} \cdot V+\frac{3 P_{0}}{2 R}$
for $\frac{d T}{d V}=0 \Rightarrow \frac{P_{0}}{R V_{0}} \cdot V=\frac{3 P_{0}}{2 R}$
$\Rightarrow V=\frac{3 V_{0}}{2}$
This is the critical point.
$\frac{d^{2} T}{d V^{2}}=-\frac{P_{0}}{R V_{0}}$
which is negative since pressure and volume can't be negative.
$\Rightarrow$ pt of maximum
Now at $V=\frac{3 V_{0}}{2}$
maximum temperature $=T_{\max }=\frac{-P_{0}}{2 R V_{0}} \frac{9 V_{0}^{2}}{4}+\frac{3 P_{0}}{2 R} \cdot \frac{3 V_{0}}{2}$
$\Rightarrow T_{\max }=\frac{9 P_{0} V_{0}}{8 R}$
Download our appand get started for free
Similar Questions
- 1View SolutionThe internal energy of an ideal gas increases during an isothermal process when the gas is
- 2A sample of $1$ mole gas at temperature $\mathrm{T}$ is adiabatically expanded to double its volume. If adiabatic constant for the gas is $\gamma=\frac{3}{2}$, then the work done by the gas in the process is:View Solution
- 3A piece of hot copper at $100^{\circ} C$ is plunged into a pond at $30^{\circ} C$. The copper cools down to $30^{\circ} C$, while the pond being huge stays at its initial temperature. Then,View Solution
- 4A gas ($\gamma = 1.3)$ is enclosed in an insulated vessel fitted with insulating piston at a pressure of ${10^5}\,N/{m^2}$. On suddenly pressing the piston the volume is reduced to half the initial volume. The final pressure of the gas isView Solution
- 5Answer the following by appropriately matching the lists based on the information given in the paragraph.View Solution
In a thermodynamics process on an ideal monatomic gas, the infinitesimal heat absorbed by the gas is given by $T \Delta X$, where $T$ is temperature of the system and $\Delta X$ is the infinitesimal change in a thermodynamic quantity $X$ of the system. For a mole of monatomic ideal gas
$X=\frac{3}{2} R \ln \left(\frac{T}{T_A}\right)+R \ln \left(\frac{V}{V_A}\right)$. Here, $R$ is gas constant, $V$ is volume of gas, $T_A$ and $V_A$ are constants.
The $List-I$ below gives some quantities involved in a process and $List-II$ gives some possible values of these quantities.
List-$I$ List-$II$ $(I)$ Work done by the system in process $1 \rightarrow 2 \rightarrow 3$ $(P)$ $\frac{1}{3} R T_0 \ln 2$ $(II)$ Change in internal energy in process $1 \rightarrow 2 \rightarrow 3$ $(Q)$ $\frac{1}{3} RT _0$ $(III)$ Heat absorbed by the system in process $1 \rightarrow 2 \rightarrow 3$ $(R)$ $R T _0$ $(IV)$ Heat absorbed by the system in process $1 \rightarrow 2$ $(S)$ $\frac{4}{3} RT _0$ $(T)$ $\frac{1}{3} RT _0(3+\ln 2)$ $(U)$ $\frac{5}{6} RT _0$ If the process carried out on one mole of monatomic ideal gas is as shown in figure in the PV-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is,
$(1)$$I \rightarrow Q, II \rightarrow R , III \rightarrow P , IV \rightarrow U$
$(2)$ $I \rightarrow S , II \rightarrow R , III \rightarrow Q , IV \rightarrow T$
$(3)$ $I \rightarrow Q , II \rightarrow R , III \rightarrow S , IV \rightarrow U$
$(4)$ $I \rightarrow Q , II \rightarrow S , III \rightarrow R , IV \rightarrow U$
($2$) If the process on one mole of monatomic ideal gas is an shown is as shown in the $TV$-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is
$(1)$ $I \rightarrow S, II \rightarrow T, III \rightarrow Q , IV \rightarrow U$
$(2)$ $I \rightarrow P , II \rightarrow R, III \rightarrow T , IV \rightarrow S$
$(3)$ $I \rightarrow P, II \rightarrow, III \rightarrow Q, IV \rightarrow T$
$(4)$ $I \rightarrow P, II \rightarrow R, III \rightarrow T, IV \rightarrow P$
Give the answer or quetion $(1)$ and $(2)$
- 6The pressure and volume of an ideal gas are related as $\mathrm{PV}^{3 / 2}=\mathrm{K}$ (Constant). The work done when the gas is taken from state $A\left(P_1, V_1, T_1\right)$ to state $\mathrm{B}\left(\mathrm{P}_2, \mathrm{~V}_2, \mathrm{~T}_2\right)$ is :View Solution
- 7A long cylindrical pipe of radius $20 \,cm$ is closed at its upper end and has an airtight piston of negligible mass as shown. When a $50 \,kg$ mass is attached to the other end of the piston, it moves down. If the air in the enclosure is cooled from temperature $T$ to $T-\Delta T$, the piston moves back to its original position. Then $\Delta T / T$ is close to (Assuming air to be an ideal gas, $g=10 \,m / s ^2$, atmospheric pressure is $10^5 \,Pa$ )View Solution
- 8View Solution“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
- 9Half mole of an ideal monoatomic gas is heated at constant pressure of $1\, atm$ from $20\,^oC$ to $90\,^oC$. Work done by has is close to ..... $J$ (Gas constant $R = 8.31\, J/mol.K$)View Solution
- 10A Container having $1$ mole of a gas at a temperature $27°C$ has a movable piston which maintains at constant pressure in container of $1 \,atm.$ The gas is compressed until temperature becomes $127°C$. The work done is ........ $J$ ($C_P$ for gas is $7.03\, cal/mol-K)$View Solution
