Two gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally
Advanced
Work done is equal to area under the P-V graph, thus work done by gas is greater for isothermal process.
For isothermal process: $T_{2}=T_{o}$
For adibatic process: $T_{o}\left(V_{o}\right)^{\gamma-1}=T(V)^{\gamma-1}$
As $V>V_{o} \Longrightarrow T Thus final temp. is greater for isothermal process. For isothermal process: $P_{o} V_{o}=P_{i} V \quad \Longrightarrow P_{i}=P_{o} \frac{V_{o}}{V}$ For adibatic process: $P_{o}\left(V_{o}\right)^{\gamma}=P_{a}(V)^{\gamma}$ $\Longrightarrow P_{a}=P_{o}\left(\frac{V_{o}}{V}\right)^{\gamma}$ As $\gamma>1 \quad$ (always) $\Longrightarrow P_{i}>P_{a}$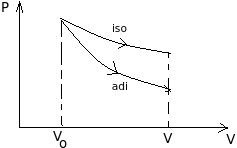
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A cyclic process $ABCA$ is shown in $PT$ diagram. When presented on $PV$, it wouldView Solution
- 2An ideal gas is made to undergo a cycle depicted by the $p-V$ diagram given below. The curved line from $A$ to $B$ is an adiabat.Then,View Solution
- 3Half mole of an ideal monoatomic gas is heated at constant pressure of $1\, atm$ from $20\,^oC$ to $90\,^oC$. Work done by has is close to ..... $J$ (Gas constant $R = 8.31\, J/mol.K$)View Solution
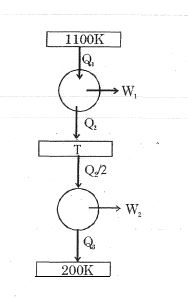
- 4Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
- 5View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 6Column $I$ Contains a list of processes involving expansion of an ideal gas. Match this with Column $II$ describing the thermodynamic change during this process. Indicate your answer by darkening the appropriate bubbles of the $4 \times 4$ matrix given in the $ORS$.View Solution
Column $I$ Column $II$ $(A)$ An insulated container has two chambers separated by a valve. Chamber $I$ contains an ideal gas and the Chamber $II$ has vacuum. The valve is opened. $(p)$ The temperature of the gas decreases $(B)$ An ideal monoatomic gas expands to twice its original volume such that its pressure $\mathrm{P} \propto \frac{1}{\mathrm{~V}^2}$, where $\mathrm{V}$ is the volume of the gas $(q)$ The temperature of the gas increases or remains constant $(C)$ An ideal monoatomic gas expands to twice its original volume such that its pressure $\mathrm{P} \propto \frac{1}{\mathrm{~V}^{4 / 3}}$, where $\mathrm{V}$ is its volume $(r)$ The gas loses heat $(D)$ An ideal monoatomic gas expands such that its pressure $\mathrm{P}$ and volume $\mathrm{V}$ follows the behaviour shown in the graph $Image$ $(s)$ The gas gains heat 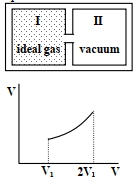
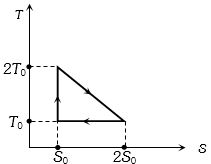
- 7A cyclic process $ABCA$ is shown in the $V-T$ diagram. Process on the $P-V$ diagram isView Solution
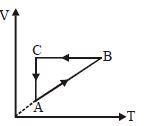
- 8Three moles of an ideal gas $\left( {{C_P} = \frac{7}{2}R} \right)$ at pressure ${P_A}$ and temperature ${T_A}0$ is isothermally expanded to twice its initial volume. It is then compressed at constant pressure to its original volume. Finally the gas is compressed at constant volume to its original pressure ${P_A}.$ The correct $P-V$ and $P-T$ diagrams indicating the process areView Solution
- 9An ideal gas goes from state $A$ to state $B$ via three different processes as indicated in the $P-V$ diagram. If $Q_1, Q_2, Q_3$ indicate the heat absorbed by the gas along the three processes and $\Delta U_1, \Delta U_2, \Delta U_3$View Solution
indicate the change in internal energy along the three processes respectively, then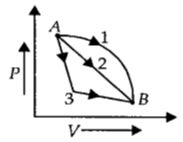
- 10View SolutionThe internal energy of the gas increases In
