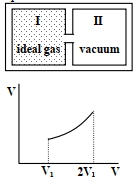
Which of the following statement($s$) is(are) correct?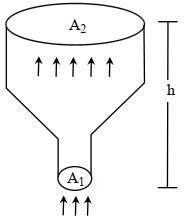
$v _1=\frac{0.8}{0.2 \times 0.1}=40 m / s$
$g=10 m / s ^2$
$\gamma=2$
Gas undergoes adiabatic expansion,
$p ^{1 \gamma} T ^\gamma=\text { Constant }$
$\frac{ P _2}{ P _1}=\left(\frac{ T _1}{ T _2}\right)^{\frac{ T }{1-\gamma}}$
$P _2=\left(\frac{300}{150}\right)^{\frac{2}{-1}} \times 600$
$P _2=\frac{600}{4}=150 Pa$
Now $\rho=\frac{ PM }{ RT } \Rightarrow \rho \propto \frac{ P }{ T }$
$\frac{\rho_1}{\rho_2}=\left(\frac{ P _1}{ P _2}\right)\left(\frac{ T _1}{ T _2}\right)=\left(\frac{150}{600}\right)\left(\frac{300}{150}\right)=\frac{1}{2}$
$\rho_2=\frac{\rho_1}{2}=0.1 kg / m ^3$
$\text { Now } \rho_2 A _2 V _2=0.8 \Rightarrow v _2=\frac{0.8}{0.1 \times 0.4}=20 m / s$
$\text { Now } W _{\text {on gas }}=\Delta K +\Delta U +\text { (Internal energy) }$
$P _1 A _1 \Delta x _1- P _2 A _2 \Delta x _2=\frac{1}{2} \Delta mV_{2 } ^ { 2 } -\frac{1}{2} \Delta mV V _1^2+\Delta mgh +\frac{ f }{2}\left( P _2 \Delta V _2- P _1 \Delta V _1\right)$
$\Rightarrow 2 P _1 \frac{\Delta V _1}{\Delta m }-2 P _2 \frac{\Delta V _2}{\Delta m }=\frac{ V _2^2- V _1^2}{2}+ gh$
$\Rightarrow \frac{2 \times 600}{0.2}-\frac{2 \times 150}{0.1}=\frac{20^2-40^2}{2}+10 h$
$h =360 m$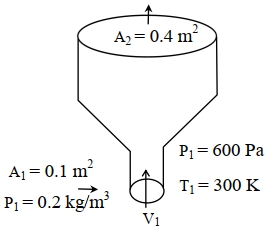
Download our appand get started for free
Similar Questions
- 1Suppose ideal gas equation follows $V{P^3}$= constant. Initial temperature and volume of the gas are $T$ and $V$ respectively. If gas expand to $27V$ then its temperature will be comeView Solution
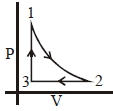
- 2Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
- 3$2$ moles of a monoatomic gas are expanded to double its initial volume, through a process $P/V =$ constant. If its initial temperature is $300\,\, K$, then which of the following is not true.View Solution
- 4View SolutionFor free expansion of the gas which of the following is true
- 5An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............View Solution
- 6An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$View Solution
- 7View SolutionConsider a process shown in the figure. During this process the work done by the system
- 8A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure isView Solution
- 9The internal energy change in a system that has absorbed $2 \;k cal$ of heat and done $500 \;J $ of work is ...... $J$View Solution
- 10Column $I$ Contains a list of processes involving expansion of an ideal gas. Match this with Column $II$ describing the thermodynamic change during this process. Indicate your answer by darkening the appropriate bubbles of the $4 \times 4$ matrix given in the $ORS$.View Solution
Column $I$ Column $II$ $(A)$ An insulated container has two chambers separated by a valve. Chamber $I$ contains an ideal gas and the Chamber $II$ has vacuum. The valve is opened. $(p)$ The temperature of the gas decreases $(B)$ An ideal monoatomic gas expands to twice its original volume such that its pressure $\mathrm{P} \propto \frac{1}{\mathrm{~V}^2}$, where $\mathrm{V}$ is the volume of the gas $(q)$ The temperature of the gas increases or remains constant $(C)$ An ideal monoatomic gas expands to twice its original volume such that its pressure $\mathrm{P} \propto \frac{1}{\mathrm{~V}^{4 / 3}}$, where $\mathrm{V}$ is its volume $(r)$ The gas loses heat $(D)$ An ideal monoatomic gas expands such that its pressure $\mathrm{P}$ and volume $\mathrm{V}$ follows the behaviour shown in the graph $Image$ $(s)$ The gas gains heat