Suppose ideal gas equation follows $V{P^3}$= constant. Initial temperature and volume of the gas are $T$ and $V$ respectively. If gas expand to $27V$ then its temperature will be come
Medium
(b) $V{P^3}$= constant = $k$ ==> $P = \frac{k}{{{V^{1/3}}}}$
Also$PV = \mu RT$==>$\frac{k}{{{V^{1/3}}}}.V = \mu RT$==> ${V^{2/3}} = \frac{{\mu RT}}{k}$
Hence ${\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^{2/3}} = \frac{{{T_1}}}{{{T_2}}}$==> ${\left( {\frac{V}{{27V}}} \right)^{2/3}} = \frac{T}{{{T_2}}}$==> ${T_2} = 9\,T$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
- 2A Carnot engine takes $6000 \,cal$ of heat from a reservoir at $627^{\circ} C$ and gives it to a sink at $27^{\circ} C$. The work done by the engine is ......... $kcal$View Solution
- 3One mole of a monoatomic ideal gas $\left(c_{ V }=\frac{3}{2} R \right)$ undergoes a cycle where it first goes isochorically from the state $\left(\frac{3}{2} P _0, V _0\right)$ to $\left( P _0, V _0\right)$, and then is isobarically contracted to the volume $\frac{1}{2} V _0$. It is then taken back to the initial state by a path which is a quarter ellipse on the $P - V$ diagram. The efficiency of this cycle isView Solution
- 4View SolutionFor free expansion of the gas which of the following is true
- 5A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution
- 6A hypothetical gas expands adiabatically such that its volume changes from $8$ litres to $27$ litres. If the ratio of final pressure of the gas to initial pressure of the gas is $\frac{16}{81}$. Then the ratio of $\frac{C_P}{C_V}$ will beView Solution
- 7The change in the entropy of a $1$ mole of an ideal gas which went through an isothermal process from an initial state $(P_1, V_1,T)$ to the final state $(P_2, V_2,T)$ is equal toView Solution
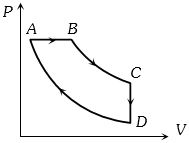
- 8The $P-V$ diagram of a certain process (carnot cycle) is as shown. The process is also represent asView Solution
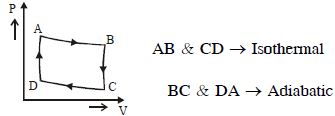
- 9Consider a carnot's cycle operating between $T_1 = 500\,K$ and $T_2 = 300\,K$ producing $1\,kJ$ of mechanical work per cycle. Find the heat transferred to the engine by the reservoirs .... $J$View Solution
- 10View SolutionIn thermodynamic processes which of the following statements is not true?
