The internal energy change in a system that has absorbed $2 \;k cal$ of heat and done $500 \;J $ of work is ...... $J$
AIPMT 2009, Medium
(a)$\Delta Q = 2k\,cal = 2 \times {10^3} \times 4.2J$$ = 8400J$ and $\Delta W = 500J.$
Hence from $\Delta Q = \Delta U + \Delta W$
$\Delta U = \Delta Q - \Delta W$
$= 8400 -500 = 7900 J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
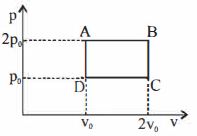
- 1The above $P-V$ diagram represents the thermodynamic cycle of an engine, operating with an ideal monatomic gas. The amount of heat, extracted from the source in a single cycle isView Solution
- 2A bubble from bottom of lake rises to its surface. Its volume doubles in the process. Assuming isothermal conditions, atmospheric pressure $= 75\, cm$ of $Hg$ and ratio of densities of mercury and water $40/3$. The depth of lake will be ..... $m$View Solution
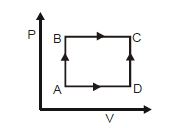
- 3A shown in the figure, the amount of heat absorbed along the path $ABC$ is $90\,J$ and the amount of work done by the system is $30\,J$ . If the amount of work done along the path $ADC$ is $20\,J$ , the amount of heat absorbed will be .... $J$View Solution
- 4At ${27^o}C$ a gas is suddenly compressed such that its pressure becomes $\frac{1}{8}th$ of original pressure. Temperature of the gas will be $(\gamma = 5/3)$View Solution
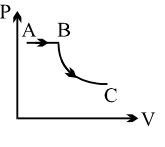
- 5View SolutionA process is shown in the diagram. Which of the following curves may represent the same process ?
- 6For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):View Solution
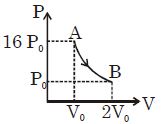
- 7Figure shows a polytropic process for an ideal gas. The work done by the gas will be in process $AB$ isView Solution
- 8An ideal gas at atmospheric pressure is adiabatically compressed so that its density becomes $32$ times of its initial value. If the final pressure of gas is $128$ atmosphers, the value of $\gamma$ the gas isView Solution
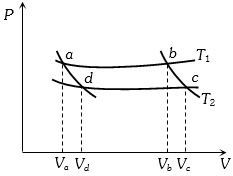
- 9In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
- 10The latent heat of vaporisation of water is $2240\, J/gm$. If the work done in the process of expansion of $1 \,g$ is $168 \,J$, then increase in internal energy is ....... $J$View Solution
