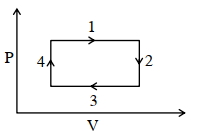
$P =$ constant, Volume increases and temperature also increases
$\Rightarrow \quad W =$ positive,$\Delta U =$ positive
$\Rightarrow \quad$ Heat is positive and supplied to gas
Process - $2$
$V =$ constant, Pressure decrease
$\Rightarrow$ Temperature decreases
$W =\int pdV =0$
$\Delta T$ is negative and $\Delta U =\frac{ f }{2} nR \Delta T$
$\Rightarrow \quad \Delta U$ in negative
$\Delta Q =\Delta U + W$
$\therefore \quad \Delta Q \rightarrow$ Heat is negative and rejected by gas
Process $3$
$P =$ constant, Volume decreases
$\Rightarrow$ Temperature also decreases
$W = P \Delta V =$ negative
$\Delta U =\frac{ f }{2} nR \Delta T =$ negative
$\Delta Q = W +\Delta U =$ negative
Heat is negative and rejected by gas.
Process $4$
$V =$ constant, Pressure increases
$W =\int pdV =0$
$PV = nRT \Rightarrow$ Temperature increase
$\Rightarrow \quad \Delta U =\frac{ f }{2} nR \Delta T$ is positive
$\Delta Q =\Delta U + W$
$=$ positive
Ans. $(C)$ step $1$ and step $4$
Download our appand get started for free
Similar Questions
- 1View SolutionCofficient of performance of refigerator is
- 2$1\, mole$ of an ideal monoatomic gas at temperature $'T_0'$ expands slowly according to the law $P = KV$, where $K$ is a constant. If the final temperature of the gas is $2T_0$. Find the heat supplied to the gasView Solution
- 3Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram.The work done on the gas in taking it from $D$ to $A$ isView Solution

- 4The efficiency of the cycle shown below in the figure (consisting of one isobar, one adiabat and one isotherm) is $50 \%$. The ratio $x$, between the highest and lowest temperatures attained in this cycle obeys (the working substance is an ideal gas)View Solution
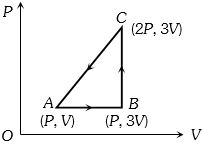
- 5An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
- 6View SolutionEntropy of a thermodynamic system does not change when this system is used for
- 7An ideal gas with constant heat capacity $C_V=\frac{3}{2} n R$ is made to carry out a cycle that is depicted by a triangle in the figure given below. The following statement is true about the cycle.View Solution
- 8Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternativeView Solution
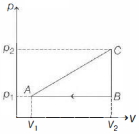
- 9For $P-V$ diagram of a thermodynamic cycle as shown in figure, process $B C$ and $D A$ are isothermal. Which of the corresponding graphs is correct?View Solution
- 10Heat given to a system is $35$ joules and work done by the system is $15$ joules. The change in the internal energy of the system will be ..... $J$View Solution
