Cofficient of performance of refigerator is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe specific heat of a gas at constant pressure is more than that of the same gas at constant volume because
- 2View SolutionIn the following indicator diagram, the net amount of work done will be
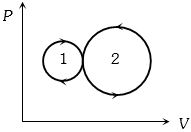
- 3An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
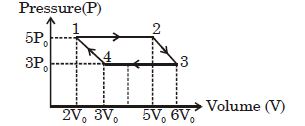
- 4A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution
- 5View SolutionThe first law of thermodynamics is concerned with the conservation of
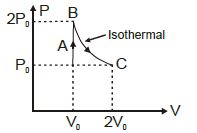
- 6View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
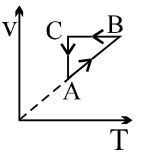
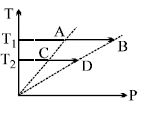
- 7On a $TP$ diagram, two moles of ideal gas perform process $AB$ and $CD$. If the work done by the gas in the process $AB$ is two times the work done in the process $CD$ then what is the value of $T_1/T_2$?View Solution
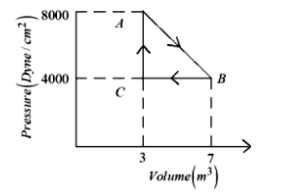
- 8A thermodynamic system is taken from an original state $A$ to an intermediate state $B$ by a linear process as shown in the figure. It's volume is then reduced to the original value from $B$ to $C$ by an isobaric process. The total work done by the gas from $A$ to $B$ and $B$ to $C$ would be :View Solution
- 9Helium at ${27^o}C$ has a volume of $8$ litres. It is suddenly compressed to a volume of $1$ litre. The temperature of the gas will be ....... $^oC$ $[\gamma = 5/3]$View Solution
- 10In an $H_2$ gas process, $PV^2 =$ constant. The ratio of work done by gas to change in its internal energy isView Solution
