Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
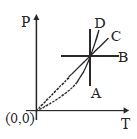
- 1View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
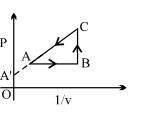
- 2One mole of a gas obeying the equation of state $P(V-b)=R T$ is made to expand from a state with coordinates $\left(P_{1}, V_{1}\right)$ to a state with $\left(P_{2}, V_{2}\right)$ along a process that is depicted by a straight line on a $P-V$ diagram. Then, the work done is given byView Solution
- 3Given below are two statements:View Solution
Statement $I:$ If heat is added to a system, its temperature must increase.
Statement $II:$ If positive work is done by a system in a thermodynamic process, its volume must increase.
In the light of the above statements, choose the correct answer from the options given below
- 4In an isothermal reversible expansion, if the volume of $96\, gm$ of oxygen at $27°C$ is increased from $70$ litres to $140$ litres, then the work done by the gas will beView Solution
- 5If a Carnot’s engine functions at source temperature $127^o C$ and at sink temperature $87^o C$, what is its efficiency ....... $\%$View Solution
- 6View SolutionThe first operation involved in a Carnot cycle is
- 7The specific heat of hydrogen gas at constant pressure is ${C_P} = 3.4 \times {10^3}cal/kg{\,^o}C$ and at constant volume is ${C_V} = 2.4 \times {10^3}cal/kg{\,^o}C.$If one kilogram hydrogen gas is heated from ${10^o}C$ to ${20^o}C$ at constant pressure, the external work done on the gas to maintain it at constant pressure isView Solution
- 8Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at ${T_1} = 800K$ and rejects to sink at ${T_2}K.$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at ${T_3} = 300K.$ If the work outputs of two engines are equal, then the value of ${T_2}$ is .... $K$View Solution
- 9In changing the state of thermodynamics from $A$ to $B$ state, the heat required is $Q$ and the work done by the system is $W.$ The change in its internal energy isView Solution
- 10View SolutionMonoatomic, diatomic and triatomic gases whose initial volume and pressure are same, are compressed till their volume becomes half the initial volume.