$\mathrm{P}_{\mathrm{i}}=\mathrm{P} \quad \mathrm{P}_{\mathrm{f}}=4 \mathrm{P}$
By gas equation we know: $-V=\frac{n R T}{P}$
$\therefore \mathrm{PV}^{4 / 3}=\mathrm{constant}$
$\Rightarrow P\left(\frac{n R T}{P}\right)^{\frac{4}{3}}=$ constant
$\Rightarrow \frac{T^{\frac{4}{3}}}{p^{\frac{1}{3}}}=$ constant
$\therefore T_{f}=\left(\frac{P_{t}}{P_{i}}\right)^{\frac{4}{3}-\frac{4}{3}} \times T_{i}=\left(\frac{4 P}{P}\right)^{\frac{1}{4}} \times 300 \mathrm{K}$
$\Rightarrow T_{f}=300 \sqrt{2} \mathrm{K}$
Download our appand get started for free
Similar Questions
- 1If ${C_V} = 4.96cal/mole\, K$, then increase in internalenergy when temperature of $2$ moles of this gas is increased from $340 K$ to $342 K$ ....... $cal$View Solution
- 2In a certain thermodynamical process, the pressure of a gas depends on its volume as $kV ^{3}$. The work done when the temperature changes from $100^{\circ} C$ to $300^{\circ} C$ will be .......... $nR$, where $n$ denotes number of moles of a gas.View Solution
- 3An ideal gas is expanding such that $\mathrm{PT}^2=$ constant. The coefficient of volume expansion of the gas isView Solution
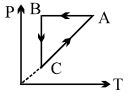
- 4A cyclic process $ABCA$ is shown in $PT$ diagram. When presented on $PV$, it wouldView Solution
- 5In thermodynamic process, $200$ Joules of heat is given to a gas and $100$ Joules of work is also done on it. The change in internal energy of the gas is ........ $J$View Solution
- 6One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $8.0 \,m ^{3}$ to $1.0 \,m ^{3}$.
Step $2$ Then expanded isothermally at temperature $T_{1}$ to volume $10.0 \,m ^{3}$.
Step $3$ Then expanded adiabatically to volume $80.0 \,m ^{3}$.
Step $4$ Then compressed isothermally at temperature $T_{2}$ to volume $8.0 \,m ^{3}$.
Then, $T_{1} / T_{2}$ is
- 7Two identical containers $A$ and $B$ with frictionless pistons contain the same ideal gas at the same temperature and the same volume $V$. The mass of the gas in $A$ is ${m_A}$ and that in $B$ is ${m_B}$. The gas in each cylinder is now allowed to expand isothermally to the same final volume $2V$. The changes in the pressure in $A$ and $B$ are found to be $\Delta P$ and $1.5 \Delta P$ respectively. ThenView Solution
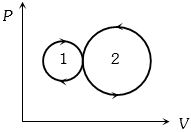
- 8View SolutionIn the following indicator diagram, the net amount of work done will be
- 9The volume of a gas is reduced adiabatically to $\frac{1}{4}$ of its volume at $27°C$, if the value of $\gamma = 1.4,$ then the new temperature will beView Solution
- 10$Assertion :$ When a bottle of cold carbonated drink is opened, a slight fog forms around the opening.View Solution
$Reason :$ Adiabatic expansion of the gas causes lowering of temperature and condensation of water vapours.
