Step $1$ It is first compressed adiabatically from volume $8.0 \,m ^{3}$ to $1.0 \,m ^{3}$.
Step $2$ Then expanded isothermally at temperature $T_{1}$ to volume $10.0 \,m ^{3}$.
Step $3$ Then expanded adiabatically to volume $80.0 \,m ^{3}$.
Step $4$ Then compressed isothermally at temperature $T_{2}$ to volume $8.0 \,m ^{3}$.
Then, $T_{1} / T_{2}$ is
Step $1 A$ to $B$ adiabatic compression, $V_{A}=8 \,m ^{3}, V_{B}=1 \,m ^{3}$
Step $2 B$ to $C$ isothermal expansion,
$T_{C}=T_{B}=T_{1}, V_{C}=10 \,m ^{3}$
Step $3 C$ to $D$ adiabatic expansion,
$V_{D}=80 \,m ^{3}$
Step $4 D$ to $A$ isothermal compression,
$T_{D}=T_{A}=T_{2}, V_{A}=8 \,m ^{3}$
Now, for processes $A$ to $B$,
$T_{A} V_{A}^{\gamma-1}=T_{B} V_{B}^{\gamma-1}$
Substituting values in above equation, we get
$T_{2}(8)^{\frac{5}{3}-1}=T_{1}(1)^{\frac{5}{3}-1}$
$\therefore$ For ideal monoatomic gas, $\gamma=\frac{5}{3}$
So, $\quad \frac{T_{1}}{T_{2}}=8^{\frac{2}{3}}=4$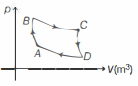
Download our appand get started for free
Similar Questions
- 1If during an adiabatic process the pressure of mixture of gases is found to be proportional to square of its absolute temperature. The ratio of $C_p / C_v$ for mixture of gases is .........View Solution
- 2View SolutionIn thermodynamic processes which of the following statements is not true?
- 3An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gasView Solution
- 4View SolutionAt constant pressure how much fraction of heat supplied to gas is converted into mechanical work?
- 5A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure isView Solution
- 6The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done by the system in going from $A \to B \to C$ is $30J$ and $40J$ heat is given to the system. The change in internal energy between $A$ and $C$ is ....... $J$View Solution
- 7A Carnot engine works between $27^{\circ} C$ and $127^{\circ} C$. Heat supplied by the source is $500\, J$. then heat ejected to the sink is (in $J$)View Solution
- 8A refrigerator consumes an average $35\, {W}$ power to operate between temperature $-10^{\circ} {C}$ to $25^{\circ} {C}$. If there is no loss of energy then how much average heat per second does it transfer? (in ${J} / {s}$)View Solution
- 9View SolutionIf a gas is heated at constant pressure, its isothermal compressibility
- 10A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
