Two identical containers $A$ and $B$ with frictionless pistons contain the same ideal gas at the same temperature and the same volume $V$. The mass of the gas in $A$ is ${m_A}$ and that in $B$ is ${m_B}$. The gas in each cylinder is now allowed to expand isothermally to the same final volume $2V$. The changes in the pressure in $A$ and $B$ are found to be $\Delta P$ and $1.5 \Delta P$ respectively. Then
IIT 1998,AIIMS 2010, Diffcult
(c) Process is isothermal. There fore, $T =$ constant,
$\left( {P \propto \frac{1}{V}} \right)$ volume is increasing, therefore pressure will decreases.
In chamber $A$ :
$\Delta P = {P_i} - {P_f} = \frac{{{\mu _A}RT}}{V} - \frac{{{\mu _A}RT}}{{2V}} = \frac{{{\mu _A}RT}}{{2V}}$…..$(i)$
In chamber $B$ :
$1.5\Delta P = {P_i} - {P_f} = \frac{{{\mu _B}RT}}{V} - \frac{{{\mu _B}RT}}{{2V}} = \frac{{{\mu _B}RT}}{{2V}}$…..$(ii)$
from equations $(i)$ and $(ii)$ $\frac{{{\mu _A}}}{{{\mu _B}}} = \frac{1}{{1.5}} = \frac{2}{3}$
==> $\frac{{{m_A}/M}}{{{m_B}/M}} = \frac{2}{3}$==> $3{m_A} = 2{m_B}.$
$\left( {P \propto \frac{1}{V}} \right)$ volume is increasing, therefore pressure will decreases.
In chamber $A$ :
$\Delta P = {P_i} - {P_f} = \frac{{{\mu _A}RT}}{V} - \frac{{{\mu _A}RT}}{{2V}} = \frac{{{\mu _A}RT}}{{2V}}$…..$(i)$
In chamber $B$ :
$1.5\Delta P = {P_i} - {P_f} = \frac{{{\mu _B}RT}}{V} - \frac{{{\mu _B}RT}}{{2V}} = \frac{{{\mu _B}RT}}{{2V}}$…..$(ii)$
from equations $(i)$ and $(ii)$ $\frac{{{\mu _A}}}{{{\mu _B}}} = \frac{1}{{1.5}} = \frac{2}{3}$
==> $\frac{{{m_A}/M}}{{{m_B}/M}} = \frac{2}{3}$==> $3{m_A} = 2{m_B}.$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
- 2For an isothermal expansion of a perfect gas, the value of $\frac{{\Delta P}}{P}$ is equalView Solution
- 3Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$View Solution
- 4In an adiabatic process, the state of a gas is changed from ${P_1},{V_1},{T_1} $ to ${P_2},{V_2},{T_2}$. Which of the following relation is correctView Solution
- 5View SolutionHeat energy absorbed by a system in going through a cyclic process shown in figure is
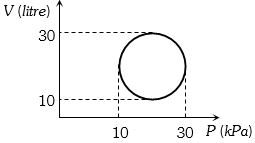
- 6View SolutionIf an ideal gas is compressed isothermally. Which of the following statements is true?
- 7One mole of a monatomic ideal gas undergoes the cyclic process $J \rightarrow K \rightarrow L \rightarrow M \rightarrow J$, as shown in the $P - T$ diagram.View Solution
Match the quantities mentioned in $List-I$ with their values in $List-II$ and choose the correct option. [ $R$ is the gas constant]
$List-I$ $List-II$ ($P$) Work done in the complete cyclic process ($1$) $R T_0-4 \ R T_0 \ln 2$ ($Q$) Change in the internal energy of the gas in the process $JK$ ($2$) $0$ ($R$) Heat given to the gas in the process $KL$ ($3$) $3 \ R T_0$ ($S$) Change in the internal energy of the gas in the process $MJ$ ($4$) $-2 \ R T_0 \ln 2$ ($5$) $-3 \ R T_0 \ln 2$ 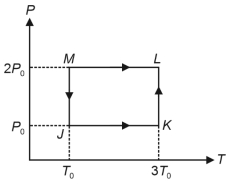
- 8View SolutionWhen an ideal monoatomic gas is heated at constant pressure, fraction of heat energy supplied which increases the internal energy of gas, is
- 9View SolutionWhen a gas expands adiabatically
- 10If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
