In thermodynamic process, $200$ Joules of heat is given to a gas and $100$ Joules of work is also done on it. The change in internal energy of the gas is ........ $J$
Easy
(b) $\Delta Q = \Delta U + \Delta W$; $\Delta Q = 200J$ and $\Delta W = - 100J$
==> $\Delta U = \Delta Q - \Delta W = 200 - ( - 100) = 300J$
==> $\Delta U = \Delta Q - \Delta W = 200 - ( - 100) = 300J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
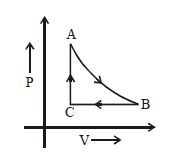
- 1The adjoining figure shows the $P-V$ diagram for a fixed mass of an ideal gas undergoing cyclic process. $AB$ represents isothermal process and $CA$ represents isochoric process.Which of the graph shown in the following figures represents the $P-T$ diagram of the cyclic process ?View Solution
- 2A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$View Solution
- 3View SolutionIn an isothermal process the volume of an ideal gas is halved. One can say that
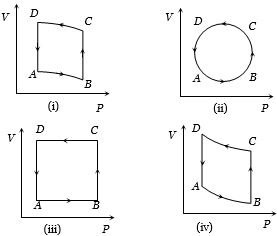
- 4In the diagrams $(i)$ to $(iv)$ of variation of volume with changing pressure is shown. A gas is taken along the path $ABCD. $ The change in internal energy of the gas will beView Solution
- 5$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
$Reason :$ Entropy increases in all natural processes. - 6The adiabatic elasticity of hydrogen gas $(\gamma = 1.4)$ at $NTP$ isView Solution
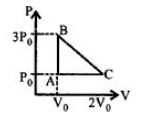
- 7One mole of an ideal monoatomic gas is taken along the path $ABCA$ as shown in the $PV$ diagram. The maximum temperature attained by the gas along the path $BC$ is given byView Solution
- 8View SolutionFor adiabatic process, wrong statement is
- 9Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
- 10View Solution“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
