In the kinetic theory of gases, which of these statements is/are true ?
$(i)$ The pressure of a gas is proportional to the mean speed of the molecules.
$(ii)$ The root mean square speed of the molecules is proportional to the pressure.
$(iii)$ The rate of diffusion is proportional to the mean speed of the molecules.
$(iv)$ The mean translational kinetic energy of a gas is proportional to its kelvin temperature.
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe ratio of specific heat of a mixture of one mole of helium and one mole of hydrogen gas will be
- 2The average translational kinetic energy of ${N}_{2}$ gas molecules at $\ldots \ldots \ldots . .{ }^{\circ} {C}$ becomes equal to the ${K} . {E}$. of an electron accelerated from rest through a potential difference of $0.1$ $volt.$View Solution
$\left(\right.$ Given $\left.{k}_{{B}}=1.38 \times 10^{-23} \, {J} / {K}\right)$
(Fill the nearest integer).
- 3View SolutionBoyle's law holds for an ideal gas during
- 4At what temperature is the kinetic energy of a gas molecule double that of its value of $27°C$View Solution
- 5One mole of ideal gas goes through process $P= 2V^2/(1+V^2)\,Pa$ then change in temperature of gas when volume changes from $V= 1\,m^3$ to $2\,m^3$ isView Solution
- 6$28\,\, gm$ of $N_2$ gas is contained in a flask at a pressure of $10$ atm and at a temperature of $57^o$. It is found that due to leakage in the flask, the pressure is reduced to half and the temperature reduced to $27\,^oC$. The quantity of $N_2$ gas that leaked out isView Solution
- 7From the following $P-T$ graph what interference can be drawnView Solution
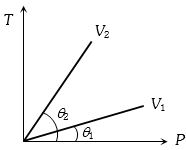
- 8View SolutionA cylinder rolls without slipping down an inclined plane, the number of degrees of freedom it has, is
- 9View SolutionThe root mean square velocity of molecules of gas is
- 10If the collision frequency of hydrogen molecules in a closed chamber at $27^{\circ} \mathrm{C}$ is $\mathrm{Z}$, then the collision frequency of the same system at $127^{\circ} \mathrm{C}$ is :View Solution
