Five particles have speeds $1, 2, 3, 4, 5 \,\,m/s$. the average velocity of the particles is (in $m/s$)
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$C{O_2}(O - C - O)$ is a triatomic gas. Mean kinetic energy of one gram gas will be (If $N-$Avogadro's number, $k-$Boltzmann's constant and molecular weight of $C{O_2} = 44$)View Solution
- 2The temperature of the hydrogen at which the average speed of its molecules is equal to that of oxygen molecules at a temperature of $31\,^oC,$ is ........ $^oC$View Solution
- 3In the kinetic theory of gases, which of these statements is/are true ?View Solution
$(i)$ The pressure of a gas is proportional to the mean speed of the molecules.
$(ii)$ The root mean square speed of the molecules is proportional to the pressure.
$(iii)$ The rate of diffusion is proportional to the mean speed of the molecules.
$(iv)$ The mean translational kinetic energy of a gas is proportional to its kelvin temperature.
- 4View SolutionThe expansion of unit mass of a perfect gas at constant pressure is shown in the diagram. Here
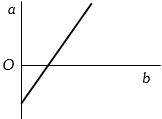
- 5View SolutionThe specific heat of a gas in a polytropic process is given by
- 6View SolutionVapour is injected at a uniform rate in a closed vessel which was initially evacuated. The pressure in the vessel
- 7The temperature $(T)$ of one mole of an ideal gas varies with its volume $(V)$ as $T=-\alpha V^3+\beta V^2$, where $\alpha$ and $\beta$ are positive constants. The maximum pressure of gas during this process is ............View Solution
- 8The root mean square speed of hydrogen molecules at $300$ $K$ is $1930\, m/s.$ Then the root mean square speed of oxygen molecules at $900\, K$ will be ....... $m/s$View Solution
- 9One mole of a monoatomic ideal gas is expanded by a process described by $p V^3=C$, where $C$ is a constant. The heat capacity of the gas during the process is given by ( $R$ is the gas constant)View Solution
- 10When water is heated from $0$ to $4\,^oC$View Solution
