An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume $V_1$ and contains ideal gas at pressure $P_1$ and temperature $T_1$ . The other chamber has volume $V_2$ and contains ideal gas at pressure $P_2$ and temperature $T_2$. If the partition is removed without doing any work on the gas, the final equilibrium temperature of the gas in the container will be
AIEEE 2008, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The average translational kinetic energy of ${O_2}$ (molar mass $32$) molecules at a particular temperature is $ 0.048\, eV.$ The translational kinetic energy of ${N_2}$ (molar mass $28$) molecules in $eV$ at the same temperature isView Solution
- 2A cubical box with porous walls containing an equal number of ${O_2}$ and $H_2$ molecules is placed in a large evacuated chamber. The entire system is maintained at constant temperature $T.$ The ratio of ${v_{rms}}$ of ${O_2}$ molecules to that of the ${v_{rms}}$ of $H_2$ molecules, found in the chamber outside the box after a short interval isView Solution
- 3The average degree of freedom per molecule of a gas is $6.$ The gas performs $25 \,J$ work, while expanding at constant pressure. The heat absorbed by the gas is ...... $J$View Solution
- 4Two non-reactive monoatomic ideal gases have their atomic masses in the ratio $2: 3$. The ratio of their partial pressures, when enclosed in a vessel kept at a constant temperature, is $4: 3$. The ratio of their densities is:View Solution
- 5The kinetic energy of one mole gas at $300K$ temperature, is $E.$ At $400K$ temperature kinetic energy is $E'.$ The value of $E'/E$ isView Solution
- 6For a diatomic gas change in internal energy for unit change in temperature for constant pressure and constant volume is ${U_1}$ and ${U_2}$ respectively. ${U_1}:{U_2}$ isView Solution
- 7The number of molecules in one litre of an ideal gas at $300 \,{K}$ and $2$ atmospheric pressure with mean kinetic energy $2 \times 10^{-9}\, {J}$ per molecules is $....\, \times 10^{11}$View Solution
- 8When the pressure on $1200\, ml$ of a gas in increased from $70\, cm $ to $120\, cm$ of mercury at constant temperature, the new volume of the gas will be ........ $ml$View Solution
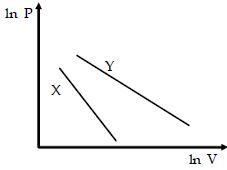
- 9For two different gases $X$ and $Y,$ having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{v_1}$ and $C_{v_2}$ respectively, for adiabatic process , the $\ln P$ versus $\ln V$ graph is plotted as shown :-View Solution
- 10$7$ mole of certain monoatomic ideal gas undergoes a temperature increase of $40 K$ at constant pressure. The increase in the internal energy of the gas in this process is$....J$ (Given $R =8.3 JK ^{-1} mol ^{-1}$ )View Solution
