The average degree of freedom per molecule of a gas is $6.$ The gas performs $25 \,J$ work, while expanding at constant pressure. The heat absorbed by the gas is ...... $J$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas initially at pressure $1$ bar is being compressed from $30 \,m ^{3}$ to $10\, m ^{3}$ volume and its temperature decreases from $320\, K$ to $280\, K$. then find final pressure of gas (in bar)View Solution
- 2A box contains n molecules of a gas. How will the pressure of the gas be effected, if the number of molecules is made $2n$View Solution
- 3Nitrogen gas is filled in an insulated container. If $\alpha$ fraction of moles dissociates without exchange of any energy, then the fractional change in its temperature is ..............View Solution
- 4At $0°C $ the density of a fixed mass of a gas divided by pressure is $x.$ At $100°C,$ the ratio will beView Solution
- 5A vertical closed cylinder is separated into two parts by a frictionless piston of mass $m$ and of negligible thickness. The piston is free to move along the length of the cylinder .The length of the cylinder above the piston is $l_1,$ and that below the piston is $l_2,$ such that $l_1 > l_2.$ Each part of the cylinder contains $n$ moles of an ideal gas at equal temperature $T.$ If the piston is stationary, its mass, $m,$ will be given by: ( $R$ is universal gas constant and $g$ is the acceleration due to gravity)View Solution
- 6The velocity of the molecules of a gas at temperature $120\,K$ is $v$. At ...... $K$ temperature will the velocity be $2v$View Solution
- 7The temperature at which the kinetic energy of oxygen molecules becomes double than its value at $27^{\circ}\,C$ is $............^{\circ}\,C$View Solution
- 8View SolutionMean kinetic energy (or average energy) per gm molecule of a monoatomic gas is given by
- 9A mixture of one mole of monoatomic gas and one mole of a diatomic gas (rigid) are kept at room temperature $\left(27^{\circ} \mathrm{C}\right)$. The ratio of specific heat of gases at constant volume respectively is:View Solution
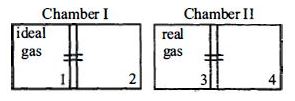
- 10There are two identical chambers, completely thermally insulated from surroundings. Both chambers have a partition wall dividing the chambers in two compartments. Compartment $1$ is filled with an ideal gas and Compartment $3$ is filled with a real gas. Compartments $2$ and $4$ are vacuum . A small hole (orifice) is made in the partition walls and the gases are allowed to expand in vacuumView Solution
Statement $-1$ : No change in the temperature of the gas takes place when ideal gas expands in vacuum. However, the temperature of real gas goes down (cooling) when it expands in vacuum
Statement $-2$ : The internal energy of an ideal gas is only kinetic. The internal energy of a real gas is kinetic as well as potential