$ = \left( {4 \times \frac{3}{2}RT + 2 \times \frac{5}{2}RT} \right) - \left( {4 \times \frac{5}{2}RT} \right) = RT$
$Note : \,(a)\, 2$ moles of diatomic gas becomes $4$ moles of a monoatomic gas when gas dissociated into atoms.
$(b)$ Internal energy of $\mu $ moles of an ideal gas of degrees of freedom $F$ is given by $U = \frac{f}{2}\mu RT$
$F = 3$ for a monoatomic gas and $5$ for diatomic gas.
Download our appand get started for free
Similar Questions
- 1Consider one mole of helium gas enclosed in a container at initial pressure $P_1$ and volume $V_1$. It expands isothermally to volume $4 V_1$. After this, the gas expands adiabatically and its volume becomes $32 V_1$. The work done by the gas during isothermal and adiabatic expansion processes are $W_{\text {iso }}$ and $W_{\text {adia, }}$ respectively. If the ratio $\frac{W_{\text {iso }}}{W_{\text {adia }}}=f \ln 2$, then $f$ is. . . . . . . .View Solution
- 2For a thermodynamic process $\delta Q = -50$ $calorie$ and $W = -20$ $calorie$ . If the initial internal energy is $-30$ $calorie$ then final internal energy will be ....... $calorie$View Solution
- 3Answer the following by appropriately matching the lists based on the information given in the paragraph.View Solution
In a thermodynamics process on an ideal monatomic gas, the infinitesimal heat absorbed by the gas is given by $T \Delta X$, where $T$ is temperature of the system and $\Delta X$ is the infinitesimal change in a thermodynamic quantity $X$ of the system. For a mole of monatomic ideal gas
$X=\frac{3}{2} R \ln \left(\frac{T}{T_A}\right)+R \ln \left(\frac{V}{V_A}\right)$. Here, $R$ is gas constant, $V$ is volume of gas, $T_A$ and $V_A$ are constants.
The $List-I$ below gives some quantities involved in a process and $List-II$ gives some possible values of these quantities.
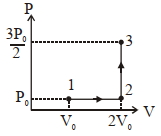
List-$I$ List-$II$ $(I)$ Work done by the system in process $1 \rightarrow 2 \rightarrow 3$ $(P)$ $\frac{1}{3} R T_0 \ln 2$ $(II)$ Change in internal energy in process $1 \rightarrow 2 \rightarrow 3$ $(Q)$ $\frac{1}{3} RT _0$ $(III)$ Heat absorbed by the system in process $1 \rightarrow 2 \rightarrow 3$ $(R)$ $R T _0$ $(IV)$ Heat absorbed by the system in process $1 \rightarrow 2$ $(S)$ $\frac{4}{3} RT _0$ $(T)$ $\frac{1}{3} RT _0(3+\ln 2)$ $(U)$ $\frac{5}{6} RT _0$ If the process carried out on one mole of monatomic ideal gas is as shown in figure in the PV-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is,
$(1)$$I \rightarrow Q, II \rightarrow R , III \rightarrow P , IV \rightarrow U$
$(2)$ $I \rightarrow S , II \rightarrow R , III \rightarrow Q , IV \rightarrow T$
$(3)$ $I \rightarrow Q , II \rightarrow R , III \rightarrow S , IV \rightarrow U$
$(4)$ $I \rightarrow Q , II \rightarrow S , III \rightarrow R , IV \rightarrow U$
($2$) If the process on one mole of monatomic ideal gas is an shown is as shown in the $TV$-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is
$(1)$ $I \rightarrow S, II \rightarrow T, III \rightarrow Q , IV \rightarrow U$
$(2)$ $I \rightarrow P , II \rightarrow R, III \rightarrow T , IV \rightarrow S$
$(3)$ $I \rightarrow P, II \rightarrow, III \rightarrow Q, IV \rightarrow T$
$(4)$ $I \rightarrow P, II \rightarrow R, III \rightarrow T, IV \rightarrow P$
Give the answer or quetion $(1)$ and $(2)$
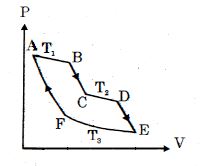
- 4One mole of ideal gas taken through a cycle process with alternate isothermal and adiabatic curves. In $P-V$ diagram $AB, CD, EF$ are isothermal curves at the absolute temperature $T_1, T_2$ and $T_3$ respectively and $BC, DE$ and $FA$ are adiabatic curves respectively. If $\frac{{{V_B}}}{{{V_A}}} = 2,\,\frac{{{V_D}}}{{{V_C}}} = 2$ then for cycle is shown in figure four statements are being made given below. (Figure is not drawn on scale)View Solution
Statement $1$ : Ratio of volumes $\frac{{{V_E}}}{{{V_F}}} = 4$
Statement $2$ : Magnitude of work done in isothermal compression $EF$ is $2RT_3\ ln\ (2)$
Statement $3$ : Ratio of heat supplied to gas in the process $AB$ to heat rejected by gas in process $EF$ is $\frac{{{T_1}}}{{2{T_3}}}$
Statement $4$ : Net work done by gas in the cycle $ABCDEFA$ is $(T_1 + T_2 - 2T_3) R\ ln\ (2)$
Find the number of correct statement $(s)$ given for the cyclic process followed by gas
- 5If heat given to a system is $6 \,kcal$ and work done is $ 6 kJ.$ Then change in internal energy is ....... $kJ$View Solution
- 6In a certain thermodynamical process, the pressure of a gas depends on its volume as $kV ^{3}$. The work done when the temperature changes from $100^{\circ} C$ to $300^{\circ} C$ will be .......... $nR$, where $n$ denotes number of moles of a gas.View Solution
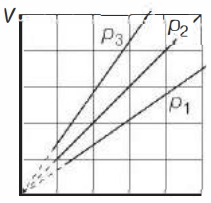
- 7View SolutionThe state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?
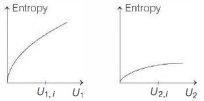
- 8Graphs below show the entropy versus energy $U$ of two systems $1$ and $2$ at constant volume. The initial energies of the systems are indicated by $U_{1, i}$ and $U_{2, i}$, respectively. Graphs are drawn to the same scale. The systems are then brought into thermal contact with each other. Assume that, at all times the combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium.View Solution
- 9A thermo-dynamical system is changed from state $({P_1},\,{V_1})$ to $({P_2},\,{V_2})$ by two different process. The quantity which will remain same will beView Solution
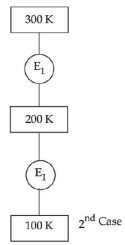
- 10In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.View Solution