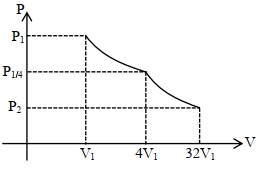
Consider one mole of helium gas enclosed in a container at initial pressure $P_1$ and volume $V_1$. It expands isothermally to volume $4 V_1$. After this, the gas expands adiabatically and its volume becomes $32 V_1$. The work done by the gas during isothermal and adiabatic expansion processes are $W_{\text {iso }}$ and $W_{\text {adia, }}$ respectively. If the ratio $\frac{W_{\text {iso }}}{W_{\text {adia }}}=f \ln 2$, then $f$ is. . . . . . . .
IIT 2020, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
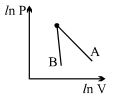
- 2The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases $A$ and $B$ undergoing adiabatic process. From figure it can be concluded thatView Solution
- 3One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
- 4A given ideal gas with $\gamma = \frac{{{C_p}}}{{{C_v}}} = 1.5$ at a temperature $T$. If the gas is compressed adiabatically to one-fourth of its initial volume, the final temperature will be ..... $T$View Solution
- 5A gas expands $0.25{m^3}$ at constant pressure ${10^3}N/{m^2}$, the work done isView Solution
- 6An electric heater supplies heat to a system at a rate of $100\;W.$ If system performs work at a rate of $75$ Joules per second. At what rate (in $J/s$) is the internal energy increasing?View Solution
- 7View SolutionIn practice, all heat engines have efficiency less than that of a Carnot engine because
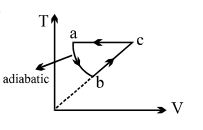
- 8$PV$ curve for the process whose $VT$ curve isView Solution
- 9View SolutionIn practice, all heat engines have efficiency less than that of a Carnot engine because
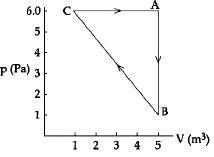
- 10For the given cyclic process $CAB$ as shown for a gas, the work done is ..... $J$View Solution