In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.
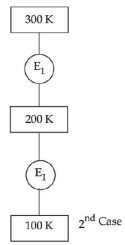

JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIf an ideal gas is compressed isothermally. Which of the following statements is true?
- 2If a Carnot’s engine functions at source temperature $127^o C$ and at sink temperature $87^o C$, what is its efficiency ....... $\%$View Solution
- 3A Carnot engine takes $3 \times {10^6}\,cal$. of heat from a reservoir at $627°C$, and gives it to a sink at $27°C.$ The work done by the engine isView Solution
- 4If $300 ml$ of a gas at ${27^o}C$ is cooled to ${7^o}C$ at constant pressure, then its final volume will be ...... $ml$View Solution
- 5The work of $146\,kJ$ is performed in order to compress one kilo mole of a gas adiabatically and in this process the temperature of the gas increases by $7\,^oC$ . The gas is $(R = 8.3\, J\, mol^{-1}\, K^{-1})$View Solution
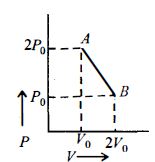
- 6$n\, moles$ of an ideal gas undergo a process $A \to B$ as shown in the figure. Maximum temperature of the gas during the process isView Solution
- 7In an adiabatic change, the pressure $P$ and temperature $T$ of a monoatomic gas are related by the relation $P \propto {T^C}$, where $C$ equalsView Solution
- 8A diatomic gas, having $C_{p}=\frac{7}{2} R$ and $C _{ v }=\frac{5}{2} R ,$ is heated at constant pressure. The ratio $dU : dQ : dW :$View Solution
- 9$1\, mole$ of an ideal monoatomic gas at temperature $'T_0'$ expands slowly according to the law $P = KV$, where $K$ is a constant. If the final temperature of the gas is $2T_0$. Find the heat supplied to the gasView Solution
- 10During which of the following thermodynamic process represented by $P V$ diagram the heat energy absorbed by system may be equal to area under $P V$ graph?View Solution
