In a thermodynamics process on an ideal monatomic gas, the infinitesimal heat absorbed by the gas is given by $T \Delta X$, where $T$ is temperature of the system and $\Delta X$ is the infinitesimal change in a thermodynamic quantity $X$ of the system. For a mole of monatomic ideal gas
$X=\frac{3}{2} R \ln \left(\frac{T}{T_A}\right)+R \ln \left(\frac{V}{V_A}\right)$. Here, $R$ is gas constant, $V$ is volume of gas, $T_A$ and $V_A$ are constants.
The $List-I$ below gives some quantities involved in a process and $List-II$ gives some possible values of these quantities.
| List-$I$ | List-$II$ |
| $(I)$ Work done by the system in process $1 \rightarrow 2 \rightarrow 3$ | $(P)$ $\frac{1}{3} R T_0 \ln 2$ |
| $(II)$ Change in internal energy in process $1 \rightarrow 2 \rightarrow 3$ | $(Q)$ $\frac{1}{3} RT _0$ |
| $(III)$ Heat absorbed by the system in process $1 \rightarrow 2 \rightarrow 3$ | $(R)$ $R T _0$ |
| $(IV)$ Heat absorbed by the system in process $1 \rightarrow 2$ | $(S)$ $\frac{4}{3} RT _0$ |
| $(T)$ $\frac{1}{3} RT _0(3+\ln 2)$ | |
| $(U)$ $\frac{5}{6} RT _0$ |
If the process carried out on one mole of monatomic ideal gas is as shown in figure in the PV-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is,
$(1)$$I \rightarrow Q, II \rightarrow R , III \rightarrow P , IV \rightarrow U$
$(2)$ $I \rightarrow S , II \rightarrow R , III \rightarrow Q , IV \rightarrow T$
$(3)$ $I \rightarrow Q , II \rightarrow R , III \rightarrow S , IV \rightarrow U$
$(4)$ $I \rightarrow Q , II \rightarrow S , III \rightarrow R , IV \rightarrow U$
($2$) If the process on one mole of monatomic ideal gas is an shown is as shown in the $TV$-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is
$(1)$ $I \rightarrow S, II \rightarrow T, III \rightarrow Q , IV \rightarrow U$
$(2)$ $I \rightarrow P , II \rightarrow R, III \rightarrow T , IV \rightarrow S$
$(3)$ $I \rightarrow P, II \rightarrow, III \rightarrow Q, IV \rightarrow T$
$(4)$ $I \rightarrow P, II \rightarrow R, III \rightarrow T, IV \rightarrow P$
Give the answer or quetion $(1)$ and $(2)$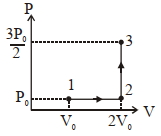
Work done in any proces $5$ Area under P-V graph $\Rightarrow$ Work dooe in $1 \rightarrow 2 \rightarrow 3= P _1 V$,
$=\frac{ RI _0}{3} \Rightarrow(Q)$
$(II)$ Change in intemal energy $1 \rightarrow 2 \rightarrow 3$
$\Delta U =\Delta C \Delta T$
$=\frac{f}{2} n R A T$
$=\frac{f}{2}\left(P_t V_t-P_2 V_i\right)$
$=\frac{3}{2}\left(\frac{3 P_0}{2} 2 V _4- P _0 V _0\right)$
$=3 P _0 V _0$
$\Delta U = RT _{ b } \Rightarrow( R )$
$(III)$ Heat absorbed in $1 \rightarrow 2 \rightarrow 3$
for any process, $I^4$ law of thermodymmics
$\Delta Q =\Delta W +\infty$
$\Delta Q = RT _4+\frac{R T_3}{3}$
$\Delta Q =\frac{4 RT }{3} \Rightarrow( s )$
$(IV)$ Heat absorbed in process $1 \rightarrow 2$
$\Delta Q=\Delta U+W$
$=\frac{f}{2}\left(P_t V_f-P_V V_0\right)+W$
$=\frac{3}{2}\left(P_a 2 V_0-P_0 V_0\right)+P_a V_0$
$=\frac{5}{2} P_0 V_a$
$=\frac{5}{2}\left(\frac{R I_0}{3}\right)$
$\Delta Q-\frac{5 R I_0}{6} \Rightarrow(U)$
($2$) Process $1 \rightarrow 2$ is isothermal (temmerature constaut)
Process $2 \rightarrow 3$ is isochoric (volme constant)
$(I)$ Work done in $1 \rightarrow 2 \rightarrow 3$
$W = W _{1 \rightarrow 2}+ W _{2 \rightarrow 1}$
$=n R T \ln \left(\frac{V_t}{V_i}\right)+ W _{2 \rightarrow 1}$
$=\frac{R I_0}{3} \ln \left(\frac{2 V_4}{V_0}\right)+0$
$W =\frac{R T_1}{3} \ln 2 \Rightarrow \text { (P) }$
$\text { (II) } \Delta U \text { in } 1 \rightarrow 2 \rightarrow 3$
$\Delta U=\frac{f}{2} n R\left(T_4-T_i\right)$
$=\frac{3}{2} R\left( T _0-\frac{T_4}{5}\right)$
$=\frac{3}{2} R\left(\frac{2 T_0}{3}\right)$
$\Delta U=R_0 \quad(R)$
$(III)$ For any system first law of thermodynamics for $1 \rightarrow 2 \rightarrow 3$
$\Delta Q=\Delta U+W$
$\Delta Q=R T_0+\frac{R T_1}{3} \ln 2$
$\Delta Q=\frac{R I_0}{3}(3+\ln 2) \Rightarrow \text { (T) }$
$(IV)$ For process $1 \rightarrow 2$ (isothermal)
$\Delta Q =\Delta U + W$
$=\frac{ f }{2} RR \left( I _e- T _{ i }\right)+ RRT \ln \left( V _e / V _{ i }\right)$
$=0+ R \left(\frac{ T _{ e }}{3}\right) \ln \left(\frac{2 v _{ e }}{ V _{ a }}\right)$
$\Delta Q =\frac{R T_4}{3} \ln 2 \Rightarrow( P )$
Download our appand get started for free
Similar Questions
- 1One mole of an ideal gas at initial temperature $T$, undergoes a quasi-static process during which the volume $V$ is doubled. During the process, the internal energy $U$ obeys the equation $U=a V^3$, where $a$ is a constant. The work done during this process isView Solution
- 2View SolutionThe efficiency of a Carnot engine depends upon
- 3An ideal gas heat engine operates in a Carnot cycle between $227^o C$ and $127^o C$. It absorbs $6\,kcal$ at the higher temperature. The amount of heat (in $kcal$) converted into work is equal toView Solution
- 4When the amount of work done is $333\, cal$ and change in internal energy is $167\, cal$, then the heat supplied is ....... $cal$View Solution
- 5An ideal gas expands in such a way that $PV^2 =$ constant throughout the process.View Solution
- 6$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
- 7A heat engine operates with the cold reservoir at temperature $324 K$. The minimum temperature of the hot reservoir, if the heat engine takes $300 \; J$ heat from the hot reservoir and delivers $180 \; J$ heat to the cold reservoir per cycle, is $\dots \; K .$View Solution
- 8Find the change in the entropy in the following process $100 \,gm$ of ice at $0°C$ melts when dropped in a bucket of water at $50°C$ (Assume temperature of water does not change) ..... $ cal/K$View Solution
- 9Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
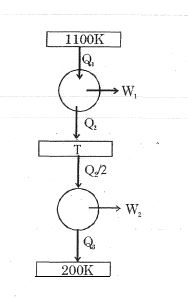
- 10A carnot engine has the same efficiency between $800 K$ to $500 K$ and $x\, K$ to $600 K.$ The value of $x$ is ...... $K$View Solution
