In a thermodynamics process on an ideal monatomic gas, the infinitesimal heat absorbed by the gas is given by $T \Delta X$, where $T$ is temperature of the system and $\Delta X$ is the infinitesimal change in a thermodynamic quantity $X$ of the system. For a mole of monatomic ideal gas
$X=\frac{3}{2} R \ln \left(\frac{T}{T_A}\right)+R \ln \left(\frac{V}{V_A}\right)$. Here, $R$ is gas constant, $V$ is volume of gas, $T_A$ and $V_A$ are constants.
The $List-I$ below gives some quantities involved in a process and $List-II$ gives some possible values of these quantities.
| List-$I$ | List-$II$ |
| $(I)$ Work done by the system in process $1 \rightarrow 2 \rightarrow 3$ | $(P)$ $\frac{1}{3} R T_0 \ln 2$ |
| $(II)$ Change in internal energy in process $1 \rightarrow 2 \rightarrow 3$ | $(Q)$ $\frac{1}{3} RT _0$ |
| $(III)$ Heat absorbed by the system in process $1 \rightarrow 2 \rightarrow 3$ | $(R)$ $R T _0$ |
| $(IV)$ Heat absorbed by the system in process $1 \rightarrow 2$ | $(S)$ $\frac{4}{3} RT _0$ |
| $(T)$ $\frac{1}{3} RT _0(3+\ln 2)$ | |
| $(U)$ $\frac{5}{6} RT _0$ |
If the process carried out on one mole of monatomic ideal gas is as shown in figure in the PV-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is,
$(1)$$I \rightarrow Q, II \rightarrow R , III \rightarrow P , IV \rightarrow U$
$(2)$ $I \rightarrow S , II \rightarrow R , III \rightarrow Q , IV \rightarrow T$
$(3)$ $I \rightarrow Q , II \rightarrow R , III \rightarrow S , IV \rightarrow U$
$(4)$ $I \rightarrow Q , II \rightarrow S , III \rightarrow R , IV \rightarrow U$
($2$) If the process on one mole of monatomic ideal gas is an shown is as shown in the $TV$-diagram with $P _0 V _0=\frac{1}{3} RT _0$, the correct match is
$(1)$ $I \rightarrow S, II \rightarrow T, III \rightarrow Q , IV \rightarrow U$
$(2)$ $I \rightarrow P , II \rightarrow R, III \rightarrow T , IV \rightarrow S$
$(3)$ $I \rightarrow P, II \rightarrow, III \rightarrow Q, IV \rightarrow T$
$(4)$ $I \rightarrow P, II \rightarrow R, III \rightarrow T, IV \rightarrow P$
Give the answer or quetion $(1)$ and $(2)$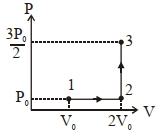
Download our appand get started for free
Similar Questions
- 1View SolutionEntropy of a system decreases .........
- 2An insulator container contains $4\, moles$ of an ideal diatomic gas at temperature $T.$ Heat $Q$ is supplied to this gas, due to which $2 \,moles$ of the gas are dissociated into atoms but temperature of the gas remains constant. ThenView Solution
- 3$2$ moles of a monoatomic gas are expanded to double its initial volume, through a process $P/V =$ constant. If its initial temperature is $300\,\, K$, then which of the following is not true.View Solution
- 4For an isothermal expansion of a perfect gas, the value of $\frac{{\Delta P}}{P}$ is equalView Solution
- 5Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 6A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
- 7The coefficient of performance of a refrigerator is $5$ . If the temperature inside freezer is $-20\,^oC$ , the temperature of the surroundings to which it rejects heat is ....... $^oC$View Solution
- 8A monoatomic gas is taken through a process $TP^{-1/3} =$ constant. If heat is given to the gasView Solution
- 9View SolutionThe slopes of isothermal and adiabatic curves are related as
- 10View SolutionWhen heat is given to a gas in an isothermal change, the result will be
