The slopes of isothermal and adiabatic curves are related as
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1When heat energy of $1500\; Joules$, is supplied to a gas at constant pressure $2.1 \times {10^5}\;N/{m^2}$, there was an increase in its volume equal to $2.5 \times {10^{ - 3}}\;{m^3}$. The increase in internal energy of the gas in Joules is ...... $J$View Solution
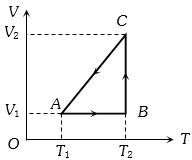
- 2A cyclic process for $1\, mole$ of an ideal gas is shown in figure in the $V-T,$ diagram. The work done in $AB, BC$ and $CA$ respectivelyView Solution
- 3View SolutionA Camot cycle consists of
- 4A gas is compressed from a volume of $2\,m^3$ to a volume of $1\, m^3$ at a constant pressure of $100\, N/m^2$. Then it is heated at constant volume by supplying $150\, J$ of energy. As a result, the internal energy of the gasView Solution
- 5Agas expands such that its initial and final temperature are equal. Also, the process followed by the gas traces a straight line on the $P-V$ diagram :View Solution
- 6View SolutionWork done on or by a gas, in general depends upon the
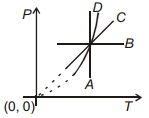
- 7Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative.View Solution
- 8A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will beView Solution
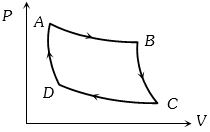
- 9The $P-V$ graph of an ideal gas cycle is shown here as below. The adiabatic process is described byView Solution
- 10Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
