Areversible adiabatic path on a $P-V$ diagram for an ideal gas passes through stateAwhere $P=0$.$7\times 10^5 \,\,N/ m^{-2}$ and $v = 0.0049 \,\,m^3$. The ratio of specific heat of the gas is $1.4$. The slope of path at $A$ is :
Diffcult
For reversible adiabat,
$P v^{\gamma}=$ constant $\Rightarrow v d P+P \gamma d v=0 \Rightarrow \frac{d P}{d v}=-\frac{\gamma P}{v}$
For $P=0.7 \times 10^{5} N m^{-2}, v=0.0049 m^{3}, \gamma=1.4$
required slope $=-\frac{1.4 \times 0.7 \times 10^{5} N m^{-2}}{0.0049 m^{3}}=-2 \times 10^{7} N m^{-5}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$200\,g$ water is heated from $40\,^oC$ to $60\,^oC.$ Ignoring the slight expansion of water, the change in its internal energy is close to ...... $kJ$ (Given specific heat of water $=4184\,J/kgK$ )View Solution
- 2The temperature of $3.00\, {mol}$ of an ideal diatomic gas is increased by $40.0^{\circ} {C}$ without changing the pressure of the gas. The molecules in the gas rotate but do not oscillate. If the ratio of change in internal energy of the gas to the amount of workdone by the gas is $\frac{{x}}{10} .$ Then the value of ${x}$ (round off to the nearest integer) is ..... . $\left(\right.$ Given $\left.{R}=8.31\, {J} {mol}^{-1} {K}^{-1}\right)$View Solution
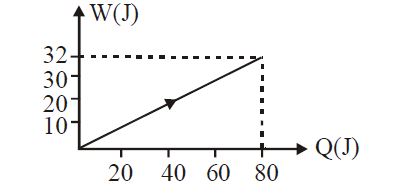
- 3View SolutionIn the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be
- 4A Carnot engine whose sink is at $300 \,K$ has an efficiency of $50 \%$. By how much should the temperature of source be increased so as the efficiency becomes $70 \%$ is ............ $K$View Solution
- 5Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at $T_1 = 800\, K$ and rejects to sink at $T_2K$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at $T_3 = 300\, K$. If the work outputs of two engines are equal, then the value of $T_2$ is ...... $K$View Solution
- 6View SolutionWhich relation is correct for isometric process
- 7Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
- 8The temperature of sink of Carnot engine is ${27^o}C$. Efficiency of engine is $25\%.$ Then temperature of source is ...... $^oC$View Solution
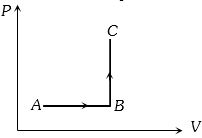
- 9The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done by the system in going from $A \to B \to C$ is $30J$ and $40J$ heat is given to the system. The change in internal energy between $A$ and $C$ is ....... $J$View Solution
- 10For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
