The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done by the system in going from $A \to B \to C$ is $30J$ and $40J$ heat is given to the system. The change in internal energy between $A$ and $C$ is ....... $J$
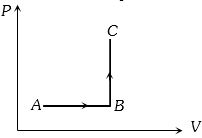

Medium
(a) Heat given $\Delta Q = 40 J$ and Work done $\Delta W = 30 J$
$⇒$ $\Delta U = \Delta Q - \Delta W = 40 -30 = 10 J.$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A hypothetical gas expands adiabatically such that its volume changes from $8$ litres to $27$ litres. If the ratio of final pressure of the gas to initial pressure of the gas is $\frac{16}{81}$. Then the ratio of $\frac{C_P}{C_V}$ will beView Solution
- 2A litre of dry air at $STP$ expands adiabatically to a volume of $3$ litres. If $\gamma=1.40,$ the work done by air is$(3^{1.4}=4.6555)$ [Take air to be an ideal gas $]$View Solution
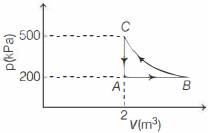
- 3A constant amount of an ideal gas undergoes the cyclic process $A B C A$ in the $p-V$ graph shown below. The path $B C$ is an isothermal. The work done by the gas during one complete cycle, beginning and ending at $A$ is nearly .......... $\,kJ$View Solution
- 4View SolutionWhen an ideal monoatomic gas is heated at constant pressure, fraction of heat energy supplied which increases the internal energy of gas, is
- 5View SolutionIn an isothermal change, an ideal gas obeys
- 6View SolutionWhich of the following process will give maximum amount of heat to surrounding when volume becomes half of initial
- 7An ideal gas is expanding such that $\mathrm{PT}^2=$ constant. The coefficient of volume expansion of the gas isView Solution
- 8Work done by a system under isothermal change from a volume ${V_1}$ to ${V_2}$ for a gas which obeys Vander Waal's equation $(V - \beta n)\,\left( {P + \frac{{\alpha {n^2}}}{V}} \right) = nRT$View Solution
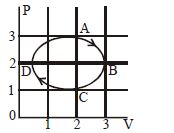
- 9The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA.$ The part $ABC$ is a semicircle and $CDA$ is half of an ellipse. Then,View Solution
- 10Heat is supplied to a diatomic gas at constant pressure. The ratio of $\Delta Q\,:\,\Delta U\,:\,\Delta W$ isView Solution
