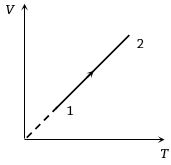
In the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
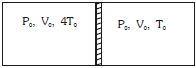
- 1Figure shows a cylindrical adiabatic container of total volume $2V_0$ divided into two equal parts by a conducting piston (which is free to move). Each part containing identical gas at pressure $P_0$ . Initially temperature of left and right part is $4T_0$ and $T_0$ respectively. An external force is applied on the piston to keep the piston at rest. Find the value of external force required when thermal equilibrium is reached. ( $A =$ Area of piston)View Solution
- 2A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
- 3Which of the following graphs correctly represents the variation of $\beta = - (dV/dP)/V$ with $P$ for an ideal gas at constant temperatureView Solution
- 4A Carnot engine takes $6000 \,cal$ of heat from a reservoir at $627^{\circ} C$ and gives it to a sink at $27^{\circ} C$. The work done by the engine is ......... $kcal$View Solution
- 5In an $H_2$ gas process, $PV^2 =$ constant. The ratio of work done by gas to change in its internal energy isView Solution
- 6A gas is suddenly compressed to $1/4$ th of its original volume at normal temperature. The increase in its temperature is ....... $K$ $(\gamma = 1.5)$View Solution
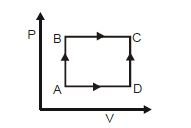
- 7A shown in the figure, the amount of heat absorbed along the path $ABC$ is $90\,J$ and the amount of work done by the system is $30\,J$ . If the amount of work done along the path $ADC$ is $20\,J$ , the amount of heat absorbed will be .... $J$View Solution
- 8$Assertion :$ Air quickly leaking out of a balloon becomes cooler.View Solution
$Reason :$ The leaking air undergoes adiabatic expansion. - 9A diatomic gas $(\gamma=1.4)$ does $400 J$ of work when it is expanded isobarically. The heat given to the gas in the process is ............ $J$View Solution
- 10Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution