Mark the correct order of decreasing acid strength of the following compounds.
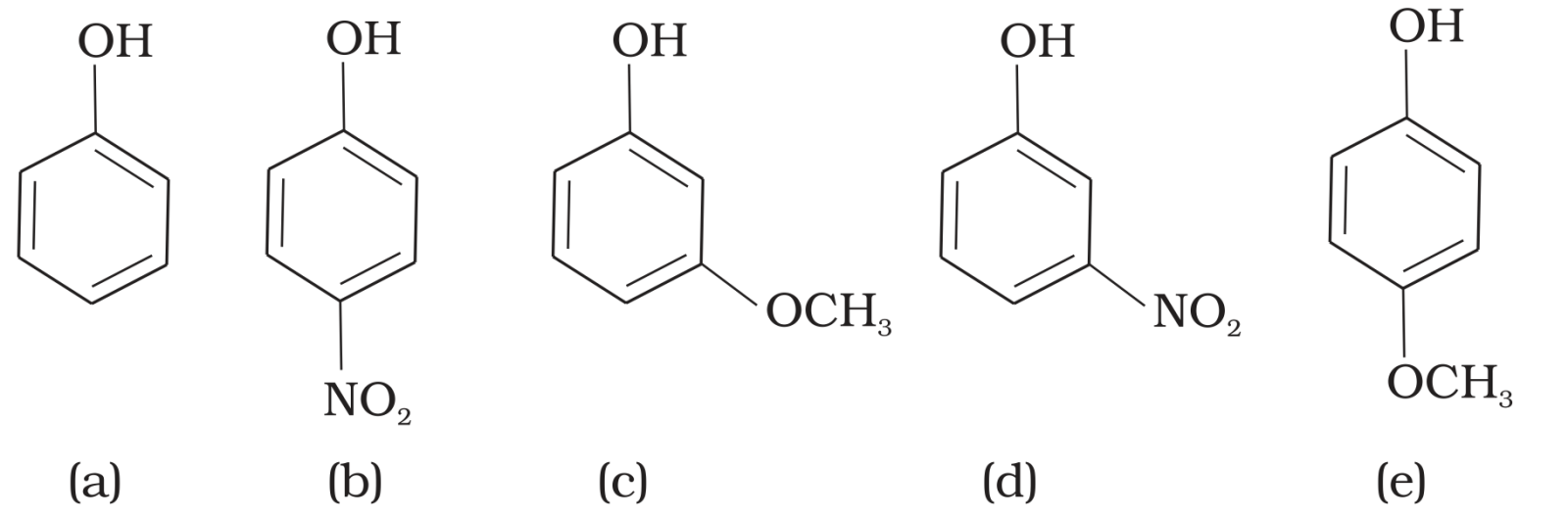

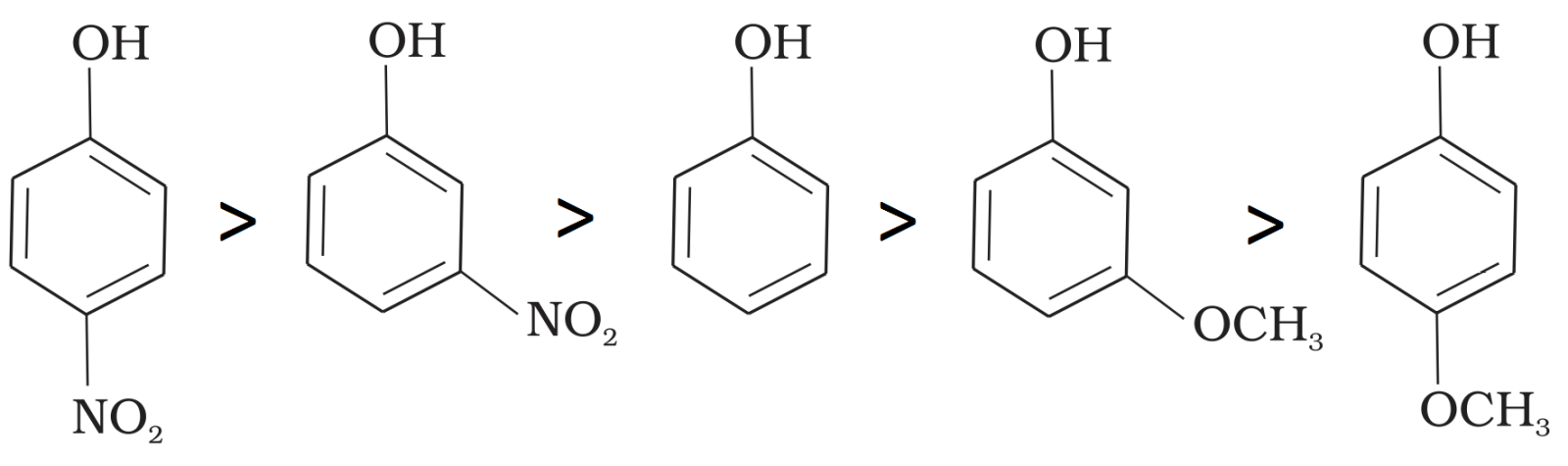
$-NO_2$ is an electron withdrawing group which increases the acidity of phenol and the effect is more pronounced at ortho and para positions. Similarly methoxy group is an electron releasing group which decreases the acidity of phenol and the effect is more pronounced at ortho and para positions.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionElectrophilic substitution reaction in phenol takes place at:
- 2View SolutionIUPAC name of phenol is -
- 3Unlike phenol, $2,4-$dinitrophenol is soluble in sodium carbonate solution in water because?View Solution
- 4View SolutionWhich of the following can not be the product of this reaction?
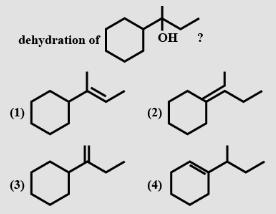
- 5View SolutionThe IUPAC name of sec. butyl alcohol is:
- 6View SolutionLower alcohols are highly soluble in water due to _____.
- 7$\ce{CH_3CH_2OH}$ can be converted into $\ce{CH_3CHO}$ by $.........$View Solution
- 8View SolutionEthers show dipolar nature due to the presence of ______.
- 9View SolutionWhich of the following compounds is aromatic alcohol?
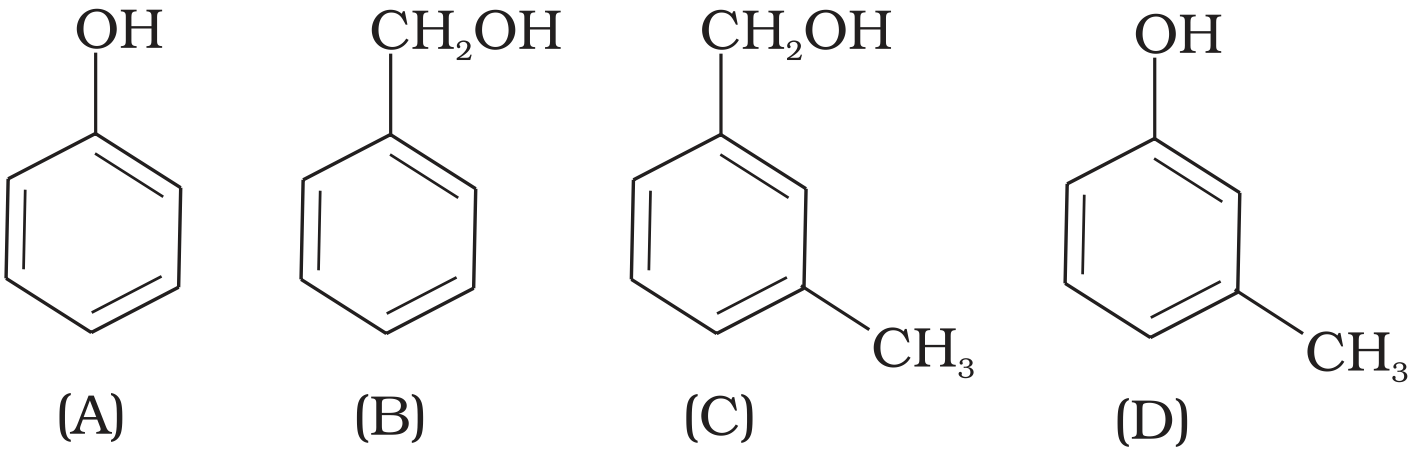
- 10View SolutionGlycol is a:
