Two ideal Carnot engines operate in cascade (all heat given up by one engine is used by the other engine to produce work) between temperatures, $\mathrm{T}_{1}$ and $\mathrm{T}_{2} .$ The temperature of the hot reservoir of the first engine is $\mathrm{T}_{1}$ and the temperature of the cold reservoir of the second engine is $\mathrm{T}_{2} . T$ is temperature of the sink of first engine which is also the source for the second engine. How is $T$ related to $\mathrm{T}_{1}$ and $\mathrm{T}_{2}$, if both the engines perform equal amount of work?
JEE MAIN 2020, Medium
$\frac{\mathrm{Q}_{\mathrm{H}}}{\mathrm{Q}_{\mathrm{L}}}=\frac{\mathrm{T}_{1}}{\mathrm{T}}$ and $\mathrm{W}=\mathrm{Q}_{\mathrm{H}}-\mathrm{Q}_{\mathrm{L}}$
$\frac{\mathrm{Q}_{\mathrm{L}}}{\mathrm{Q}_{\mathrm{L}}^{\prime}}=\frac{\mathrm{T}}{\mathrm{T}_{2}}$ and $\mathrm{W}=\mathrm{Q}_{\mathrm{L}}-\mathrm{Q}_{\mathrm{L}}$
From $(1)$ and $(2)$
we get $\mathrm{T}=\frac{\mathrm{T}_{1}+\mathrm{T}_{2}}{2}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two carnot engines $A$ and $B$ operate in series such that engine $A$ absorbs heat at $T_{1}$ and rejects heat to a sink at temperature $T$. Engine $B$ absorbs half of the heat rejected by engine $A$ and rejects heat to the sink at ${T}_{3}$. When workdone in both the cases is equal, the value of ${T}$ isView Solution
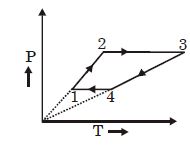
- 2Three moles of an ideal monoatomic gas perform a cycle as shown in the figure. The gas temperature in different states are: $T_1 = 400\, K, T_2 = 800\, K, T_3 = 2400\,K$ and $T_4 = 1200\,K.$ The work done by the gas during the cycle is ........ $kJ$View Solution
- 3View SolutionWhich of the following parameters does not characterize the thermodynamic state of matter
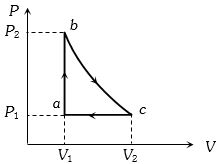
- 4Carbon monoxide is carried around a closed cycle $abc$ in which bc is an isothermal process as shown in the figure. The gas absorbs $7000 J$ of heat as its temperature increases from $300 K$ to $1000 K$ in going from $a$ to $b$. The quantity of heat rejected by the gas during the process $ca$ is ..... $J$View Solution
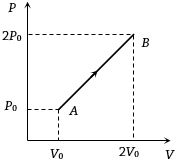
- 5The $P-V$ diagram of $2$ gm of helium gas for a certain process $A \to B$ is shown in the figure. what is the heat given to the gas during the process $A \to B$View Solution
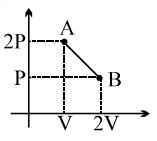
- 6One mole of a gas expands obeying the relation as shown in the $P/V$ diagram. The maximum temperature in this process is equal toView Solution
- 7View SolutionThe adiabatic Bulk modulus of a perfect gas at pressure is given by
- 8In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.View Solution
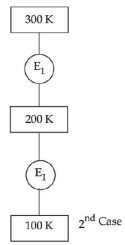
- 9The efficiency of Carnot engine when source temperature is $T_1$ and sink temperature is $T_2$ will beView Solution
- 10A total of $48 \mathrm{~J}$ heat is given to one mole of helium kept in a cylinder. The temperature of helium increases by $2^{\circ} \mathrm{C}$. The work done by the gas is : (Given, $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$.)View Solution
