At a given temperature the $r.m.s.$ velocity of molecules of the gas is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal monoatomic gas at temperature $T_0$ expands slowly according to the law $P/V$ = constant. If the final temperature is $2 \,\,T_0$, heat supplied to the gas is :View Solution
- 2A gas mixture consists of $3\,moles$ of oxygen and $5\,moles$ or argon at temperature $T.$ Considering only translational and rotational modes, the total internal energy of the system isView Solution
- 3View SolutionMean kinetic energy per degree of freedom of gas molecules is
- 4A closed vessel contains $10 g$ of an ideal gas $X$ at $300 \ K$, which exerts $2$ atm pressure. At the same temperature, $80 g$ of another ideal gas $Y$ is added to it and the pressure becomes $6 atm$. The ratio of root mean square velocities of $X$ and $Y$ at $300 \ K$ isView Solution
- 5View SolutionPressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement
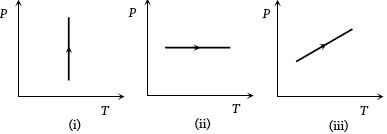
- 6View SolutionOn increasing number density for a gas in a vessel, mean free path of a gas
- 7A vessel contains $16 \,g$ of hydrogen and $128 \,g$ of oxygen at standard temperature and pressure. The volume of the vessel in $cm ^{3}$ isView Solution
- 8When heat $Q$ is supplied to a diatomic gas of rigid molecules at constant volume its temperature increases by $\Delta T.$ The heat required to produce the same change in temperature, at constant pressure isView Solution
- 9If radius of $O _{2}$ molecule $=40 \mathring A, T =27^{\circ} C$ and $P =1 \,atm .$ Find the time of relaxation.View Solution
- 10Consider two ideal diatomic gases $\mathrm{A}$ and $\mathrm{B}$ at some temperature $T$. Molecules of the gas $A$ are rigid, and have a mass $m$. Molecules of the gas $\mathrm{B}$ have an additional vibrational mode, and have a mass $\frac{\mathrm{m}}{4} .$ The ratio of the specific heats $(\mathrm{C}_{\mathrm{v}}^{\mathrm{A}}$ and $\mathrm{C}_{\mathrm{v}}^{\mathrm{B}})$ of gas $\mathrm{A}$ and $\mathrm{B}$, respectively isView Solution
