On increasing number density for a gas in a vessel, mean free path of a gas
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$7$ mole of certain monoatomic ideal gas undergoes a temperature increase of $40 K$ at constant pressure. The increase in the internal energy of the gas in this process is$....J$ (Given $R =8.3 JK ^{-1} mol ^{-1}$ )View Solution
- 2View SolutionThe specific heats at constant pressure is greater than that of the same gas at constant volume because
- 3If pressure of a gas contained in a closed vessel is increased by $0.4\%$ when heated by $1°C,$ the initial temperature must beView Solution
- 4The temperature of an ideal gas is increased from $27°C$ to $927°C.$ The root mean square speed of its molecules becomesView Solution
- 5A container has $N$ molecules at absolute temperature $T$. If the number of molecules is doubled but kinetic energy in the box remain the same as before, the absolute temperature of the gas is ...........View Solution
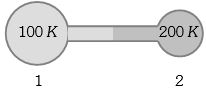
- 6Figure shows two flasks connected to each other. The volume of the flask $1$ is twice that of flask $2.$ The system is filled with an ideal gas at temperature $100\, K$ and $200 \,K $ respectively. If the mass of the gas in $1$ be $m$ then what is the mass of the gas in flask $2$View Solution
- 7View SolutionThe specific heat of a gas in a polytropic process is given by
- 8A diatomic gas of molecules weight $30\,\, gm/mole$ is filled in a container at $27\,^oC$. It is moving at a velocity $100\,\, m/s$. If it is suddenly stopped, the rise in temperature of gas is :View Solution
- 9The equation of state corresponding to $8 \,g$ of ${O_2}$ isView Solution
- 10Hydrogen gas is filled in a balloon at $20^\circ C$. If temperature is made $40^\circ C$, pressure remaining same, what fraction of Hydrogen will come outView Solution
